12.4: Aldol Addition
- Page ID
- 106367
We arrive now at one of the most important mechanisms in metabolism: the aldol addition.
Along with Claisen condensation reactions, which we will study in the next chapter, aldol additions are responsible for most of the carbon-carbon bond forming events that occur in a living cell. Because biomolecules are built upon a framework of carbon-carbon bonds, it is difficult to overstate the importance of aldol addition and Claisen condensation reactions in the chemistry of living things!
Overview of the aldol addition reaction
Consider the potential pathways available to a reactive enolate intermediate once the a-proton has been abstracted. We'll use acetaldehyde as a simple example. The oxygen, which bears most of the negative charge, could act as a base, (step 2 below) and the result would be an enol.
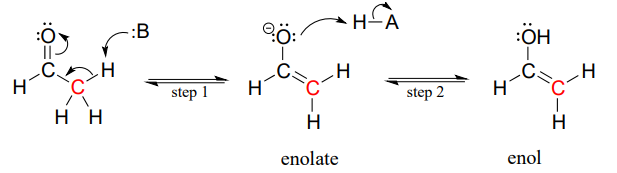
Alternatively, the enolate carbon, which bears a degree of negative charge, could act as a base, which is simply the reverse of the initial deprotonation step that formed the enolate in the first place. This of course just takes us right back to the starting aldehyde.
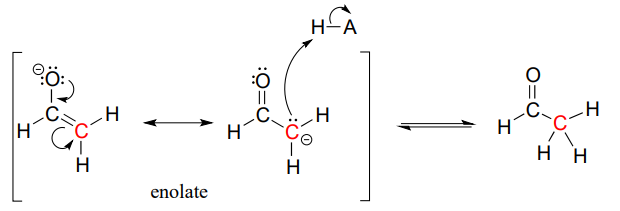
In both of these cases, the electron-poor species attacked by the enolate is an acidic proton. What if the electron-poor species - the electrophile - is not a proton but a carbonyl carbon? In other words, what if the enolate acts not as a base but rather as a nucleophile in a carbonyl addition reaction? For example, the enolate of acetaldehyde could attack the carbonyl group of a second acetaldehyde molecule. The result is the formation of a new carbon-carbon bond:

This type of reaction is called an aldol addition. It can be very helpful to think of an aldol addition reaction as simply a nucleophilic carbonyl addition (Chapter 10) reaction with an enolate a-carbon (rather than an alcohol oxygen or amine nitrogen) as the nucleophile.
An aldol addition reaction:

Mechanism:
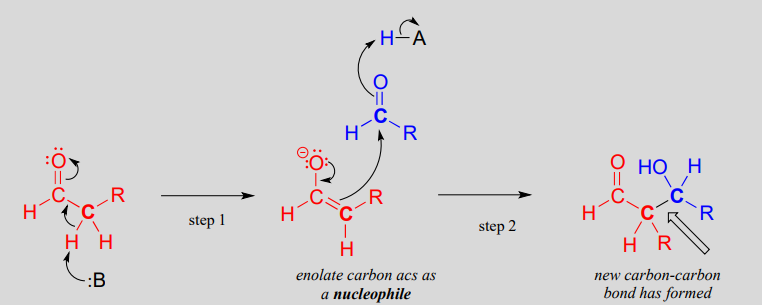
Historically, the first examples of this mechanism type to be studied involved reactions very similar to what is shown above: an aldehyde reacting with itself. Because the resulting product contained both an aldehyde and an alcohol functional group, the reaction was referred to as an 'aldol' addition, a terminology that has become standard for reactions of this type, whether or not an aldehyde is involved. More generally, an aldol addition is characterized as a nucleophilic addition to an aldehyde, ketone, or imine electrophile where the nucleophile is the a-carbon in an aldehyde, ketone, imine, ester, or thioester. The enzymes that catalyze aldol reactions are called, not surprisingly, aldolases.
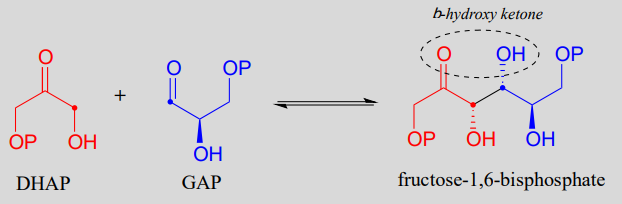
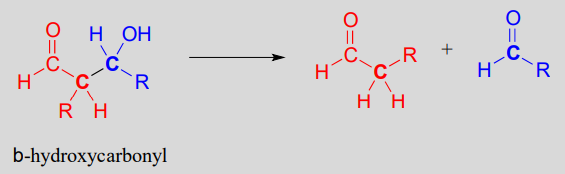
Note that the aldol reaction results in a product in which a hydroxide group is two carbons away from the carbonyl, in the \(\beta\) position. You can think of the \(\beta\)-hydroxy group as a kind of 'signature' for an aldol addition product.
Depending on the starting reactants, nonenzyatic aldol reactions can take more than one route to form different products. For example, a reaction between acetaldehyde and 2-butanone could potentially result in in three different aldol addition products, depending on which of the three a-carbons (carbons 2, 3, and 5 below) becomes the attacking nucleophile.
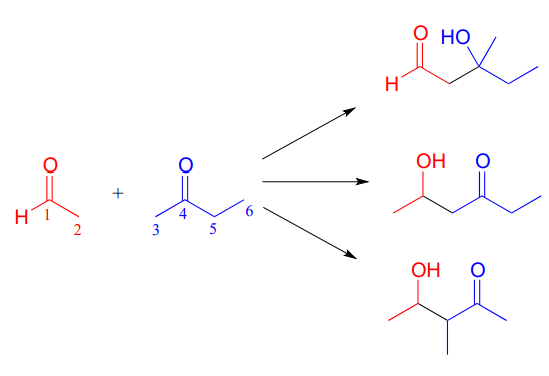
- Fill in the appropriate carbon numbers for each of the three possible aldol addition products shown above.
- Draw arrows for the carbon-carbon bond forming step that leads to each of the three products.
- Hint
-
For each reaction, first identify the nucleophilic and electrophilic carbon atoms on the starting compounds!
Biochemical aldol addition reactions
Fructose 1,6-bisphosphate aldolase (EC 4.1.2.13) is an enzyme that participates in both the glycolytic (sugar catabolism) and gluconeogenesis (sugar synthesis) biochemical pathways. The reaction catalyzed by fructose 1,6-bisphosphate aldolase links two 3-carbon sugars, glyceraldehyde-3-phosphate (GAP, the electrophile in the reaction) and dihydroxyacetone phosphate (DHAP, the nucleophile), forming a 6-carbon product. In the figures below, the nucleophilic and electrophilic carbons are identified with dots.
The fructose 1,6-bisphosphate aldolase reaction
Mechanism:
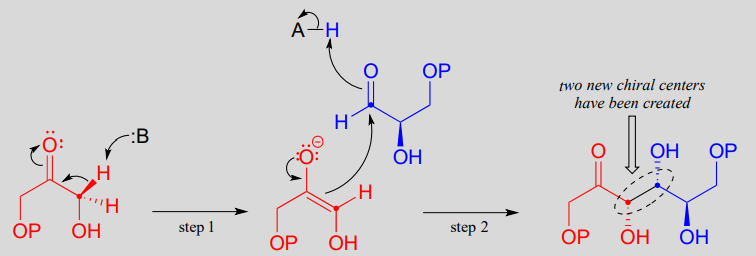
In step 1 of the reaction, an a-carbon on DHAP is deprotonated, leading to an enolate intermediate. this and many other aldolase reactions, a zinc cation (\(Zn^{+2}\)) is positioned in the enzyme's active site so as to interact closely with - and stabilize - the negatively charged oxygen of the enolate intermediate. This is one important way in which the enzyme lowers the energy barrier to the reaction.
Next, (step 2), the deprotonated a-carbon attacks the carbonyl carbon of GAP in a nucleophilic addition reaction, leading to the fructose 1,6-bisphosphate product.
Notice that two new chiral centers are created in this reaction. This reaction, being enzyme-catalyzed, is highly stereoselective due to the precise position of the two substrates in the active site: only one of the four possible stereoisomeric products is observed. The enzyme also exhibits tight control of regiochemistry: GAP and DHAP could potentially form two other aldol products which are constitutional isomers of fructose 1,6-bisposphate.
- Fill in the blanks with the correct term: (pro-R, pro-S, re, si). You may want to review the terminology in section 3.11.
In the fructose 1,6-bisphosphate aldolase reaction, the ______ proton on the a-carbon of DHAP is abstracted, then the ______ face of the resulting enolate a-carbon attacks the ______ face of the aldehyde carbon of GAP.
- Draw structures of the two other constitutional isomers that could hypothetically form in aldol addition reactions between GAP and DHAP. How many stereoisomers exist for these two alternative products?
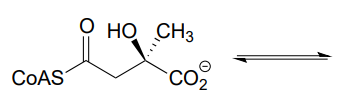
Along with aldehydes and ketones, esters and thioesters can also act as the nucleophilic partners in aldol reactions. In the first step of the citric acid (Krebs) cycle, acetyl \(CoA\) (a thioester nucleophile) adds to oxaloacetate (a ketone electrophile) (EC 2.3.3.8).
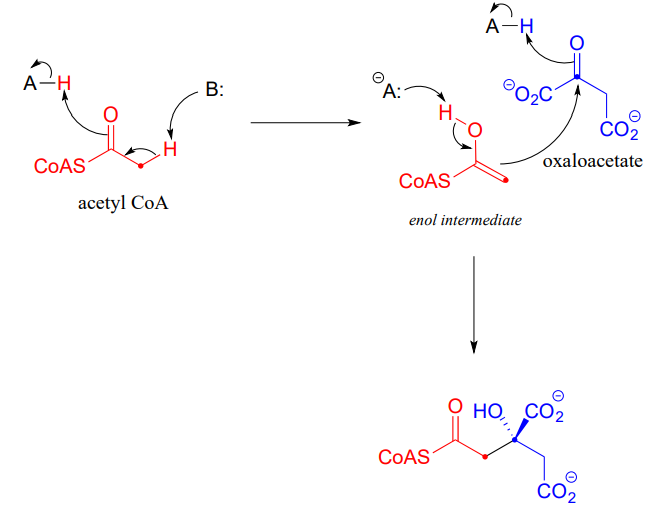
Notice that the nucleophilic intermediate is an enol, rather than a zinc-stabilized enolate as was the case with the fructose 1,6-bisphosphate aldolase reaction. An enol intermediate is often observed when the nucleophilic substrate is a thioester rather than a ketone or aldehyde.
Going backwards: the retro-aldol cleavage reaction
Although aldol reactions play a very important role in the formation of new carbon-carbon bonds in metabolic pathways, it is important to emphasize that they can also be reversible: in most cases, the energy level of starting compounds and products are very close. This means that, depending on metabolic conditions, aldolases can also catalyze retro-aldol reactions: the reverse of aldol reactions, in which carbon-carbon bonds are broken.
A retro-aldol cleavage reaction:
Mechanism:
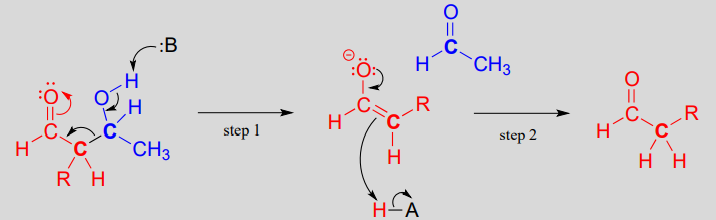
In the retro-aldol cleavage reaction the \(\beta\)-hydroxy group is deprotonated (step 1 above), to form a carbonyl, at the same time pushing off the enolate carbon, which is now a leaving group rather than a nucleophile.
Is an enolate a good enough leaving group for this step to be chemically reasonable? Sure it is: the same stabilizing factors that explain why it can form as an intermediate in the forward direction (resonance delocalization of the negative charge to the oxygen, interaction with a zinc cation) also explain why it is a relatively weak base, and therefore a relatively good leaving group (remember, weak base = good leaving group!). All we need to do to finish the reaction off is reprotonate the enolate (step 2) to yield the starting aldehyde, and we are back where we started.
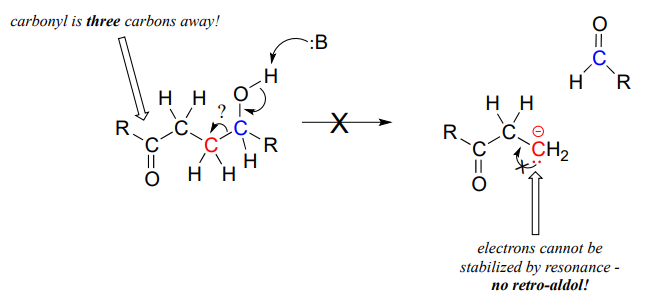
The key thing to keep in mind when looking at a retro-aldol mechanism is that, when the carbon-carbon bond breaks, the electrons must have 'some place to go' where they will be stabilized by resonance. Most often, the substrate for a retro-aldol reaction is a \(\beta\)-hydroxy aldehyde, ketone, ester, or thioester.
If the leaving electrons cannot be stabilized, a retro-aldol cleavage step is highly unlikely.
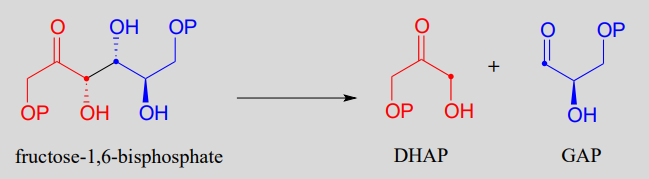
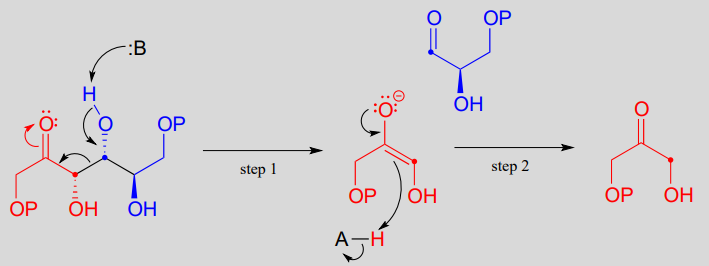
The fructose 1,6-bisphosphate aldolase reaction we saw in the previous section is an excellent example of an enzyme whose metabolic role is to catalyze both the forward and reverse (retro) directions of an aldol reaction. The same enzyme participates both as an aldolase in the sugar-building gluconeogenesis pathway, and as a retro-aldolase in the sugar breaking glycolysis pathway. We have already seen it in action as an aldolase in the gluconeogenesis pathway. Here it is in the glycolytic direction, catalyzing the retro-aldol cleavage of fructose bisphosphate into DHAP and GAP:
The fructose 1,6-bisphosphate aldolase reaction (retro-aldol direction)
Mechanism:
Predict the products of a retro-aldol reaction with the given substrate.
Aldol addition reactions with enzyme-linked enamine intermediates
Earlier we looked at the mechanism for the fructose 1,6-bisphosphate aldolase reaction in bacteria. Interestingly, it appears that the enzyme catalyzing the exact same reaction in plants and animals evolved differently: instead of going through a zinc-stabilized enolate intermediate, in plants and animals the key intermediate is an enamine. The nucleophilic substrate is first linked to the enzyme through the formation of an iminium with a lysine residue in the enzyme's active site (refer to section 10.5 for the mechanism of iminium formation). This effectively forms an 'electron sink', in which the positively-charged iminium nitrogen plays the same role as the \(Zn^{+2}\) ion in the bacterial enzyme.
Mechanism for an aldol addition reaction with an enzyme-linked enamine intermediate
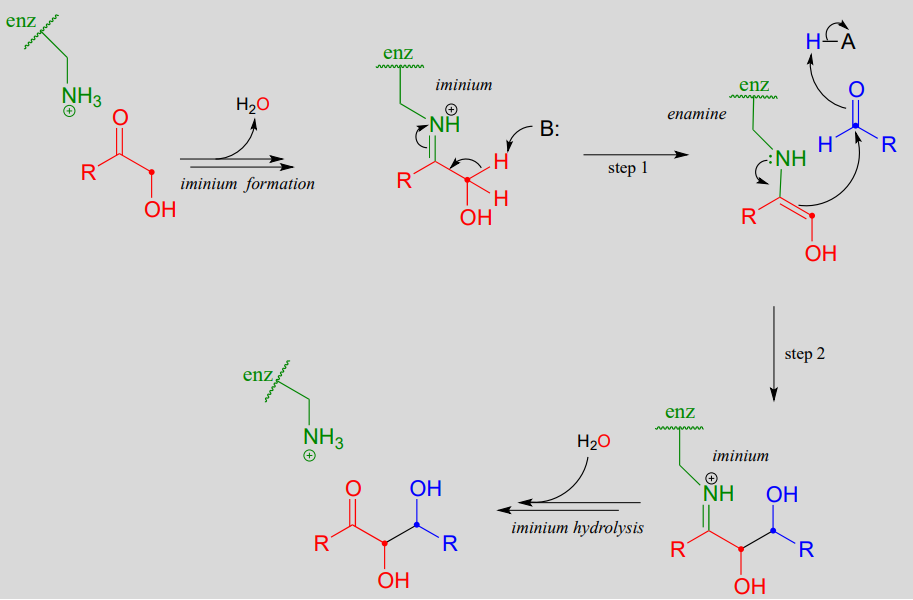
The \(\alpha\)-proton, made more acidic by the electron-withdrawing effect of the imminium nitrogen, is then abstracted by an active site base to form an enamine (step 1). In step 2 , the \(\alpha\)-carbon attacks the carbonyl carbon of an aldehyde, and the new carbon-carbon bond is formed. In order to release the product from the enzyme active site and free the enzyme to catalyze another reaction, the iminium is hydrolyzed back to a ketone group (see section 10.5 to review the imine/imminium hydrolysis mechanism).
There are many more examples of aldol/retroaldol reactions in which the key intermediate is a lysine-linked imine. Many bacteria are able to incorporate formaldehyde, a toxic compound, into carbohydrate metabolism by linking it to ribulose monophosphate. The reaction (EC 4.1.2.43) proceeds through imine and enamine intermediates.
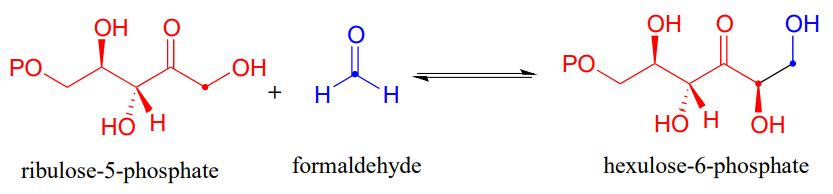
Draw the carbon-carbon bond-forming step for the hexulose-6-phosphate aldolase reaction shown above.
Here is an example of an enamine intermediate retro-aldol reaction from bacterial carbohydrate metabolism (EC 4.1.2.14). Notice that the structures are drawn here in the Fischer projection notation - it is important to practice working with this drawing convention, as biologists and biochemists use it extensively to illustrate carbohydrate chemistry. Proc. Natl. Acad. Sci. 2001, 98, 3679
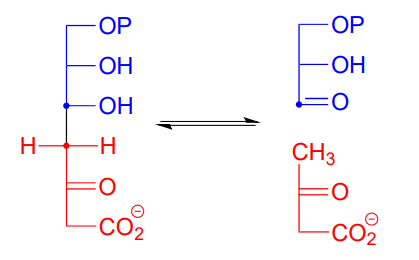
Draw the carbon-carbon bond breaking step in the reaction above. Use the Fischer projection notation.


