8.6: Epoxides as electrophiles in nucleophilic substitution reactions
- Page ID
- 1087
8.6A: Epoxide structure
Epoxides (also known as oxiranes) are three-membered ring structures in which one of the vertices is an oxygen and the other two are carbons.
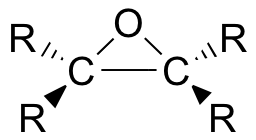
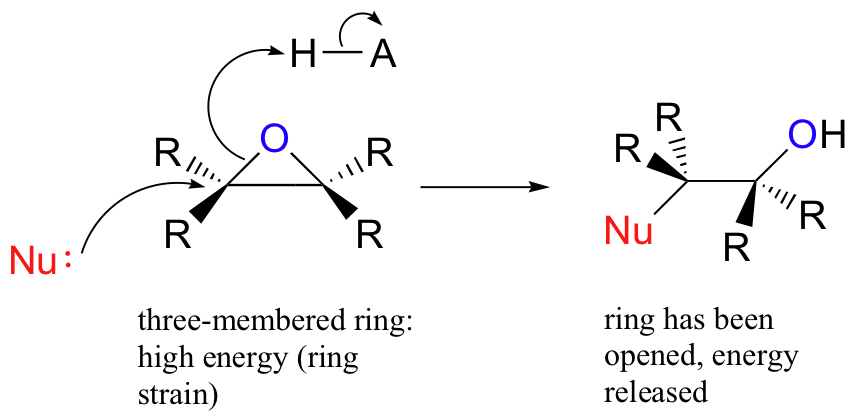
The carbons in an epoxide group are very reactive electrophiles, due in large part to the fact that substantial ring strain is relieved when the ring opens upon nucleophilic attack.
Epoxides are very important intermediates in laboratory organic synthesis, and are also found as intermediate products in some biosynthetic pathways. The compound (3S)-2,3-oxidosqualene, for example, is an important intermediate in the biosynthesis of cholesterol (we’ll see the epoxide ring-opening step in chapter 15):

Both in the laboratory and in the cell, epoxides are usually formed by the oxidation of an alkene. These reactions will be discussed in detail in chapter 16.
Exercise 8.17: Predict the major product(s) of the ring opening reaction that occurs when the epoxide shown below is treated with:
a) ethanol and a small amount of sodium hydroxide
b) ethanol and a small amount of sulfuric acid
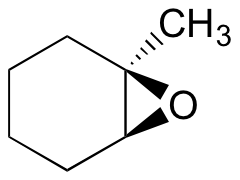
Hint: be sure to consider both regiochemistry and stereochemistry!


