6.4B: Flowcharts
- Page ID
- 95740
In some teaching labs, a combination of spectroscopy and chemical tests are used in determination of an unknown. If available, an infrared spectrometer (Figure 6.39) is very useful in determining possible functional groups present in an unknown. The following flowcharts summarize key signals present in an IR spectrum, and chemical tests that can be used to support or narrow down structural identification.

1. \(\ce{O-H}\) Bond
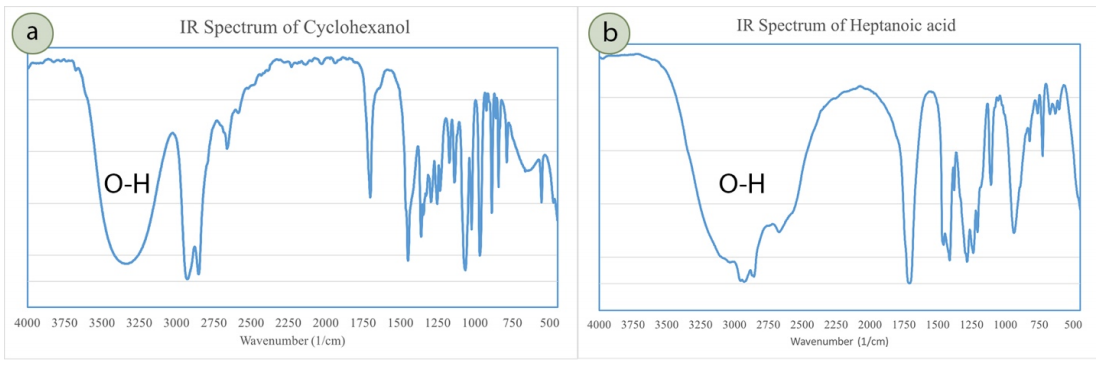
An \(\ce{O-H}\) bond strongly absorbs infrared radiation over a broad range of wavenumbers (\(2400\)-\(3400 \: \text{cm}^{-1}\)), and has a characteristic shape in IR spectra. The specific range of absorption can be used to identify whether the \(\ce{O-H}\) is part of an alcohol or carboxylic acid functional group: a broad absorption centering around \(3300 \text{cm}^{-1}\) corresponds to an alcoholic \(\ce{O-H}\) bond (Figure 6.40a), while a broader absorption centering around \(3000 \: \text{cm}^{-1}\) corresponds to a carboxylic acid \(\ce{O-H}\) bond (Figure 6.40b).
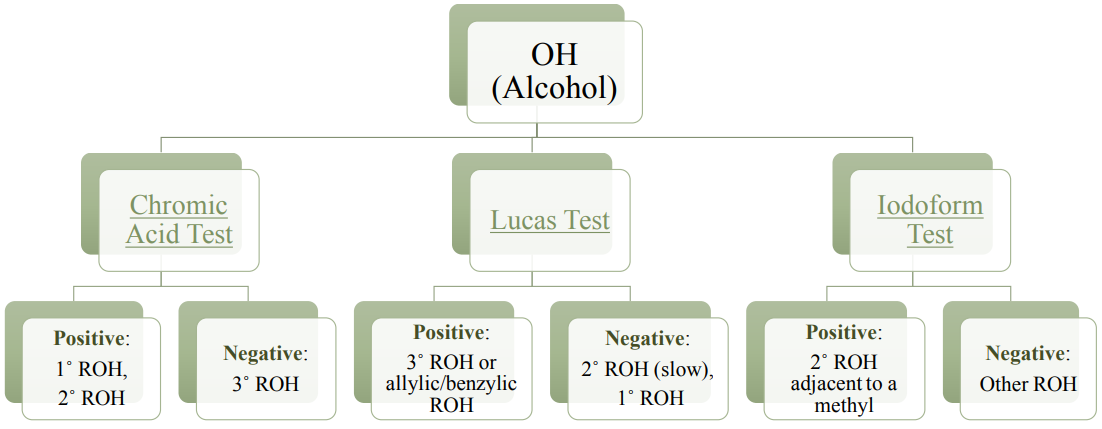
Chemical tests (pH and bicarbonate tests) can be done to support the identification of a carboxylic acid. Specific structural features of alcohols (primary, secondary, or tertiary) can be determined using a variety of chemical tests, as summarized by the flowchart in Figure 6.41.
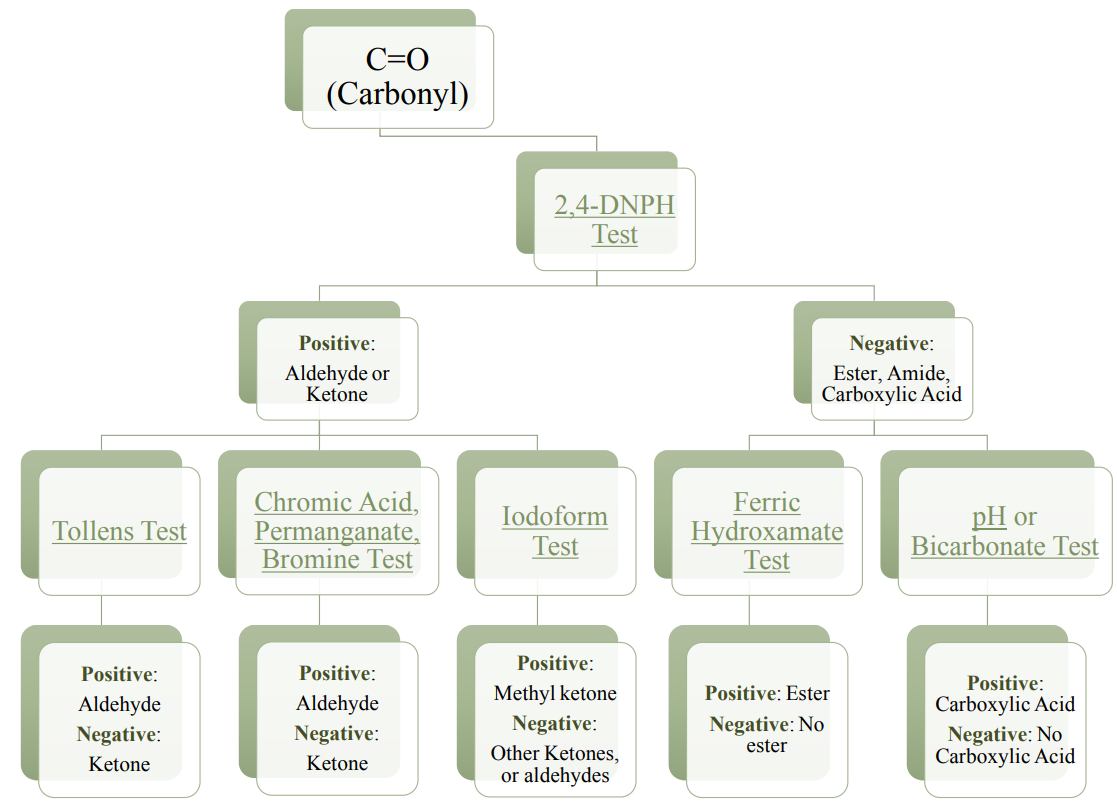
2. Carbonyl, \(\ce{C=O}\) Bond
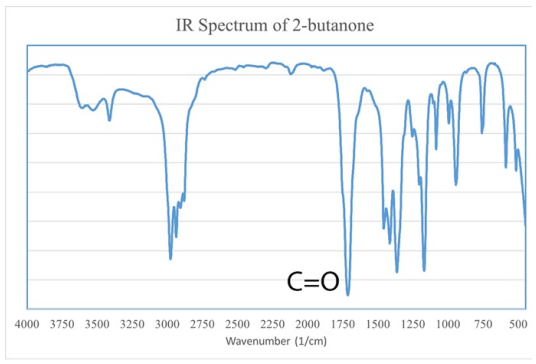
The carbonyl bond absorbs infrared radiation very strongly and sharply in the \(1700 \: \text{cm}^{-1}\) region (Figure 6.42). The specific wavenumber (\(1715 \: \text{cm}^{-1}\) or \(1735 \: \text{cm}^{-1}\) for example) often correlates well to a specific functional group (aldehyde, ketone, ester, amide, or carboxylic acid), but it is not uncommon for compounds to absorb outside of their "expected" range. Chemical tests can be useful in narrowing down the specific functional group once a carbonyl bond is identified in an IR spectrum, as summarized by the flowchart in Figure 6.43.
3. Unsaturated Hydrocarbons (Alkenes, Alkynes, Aromatics)
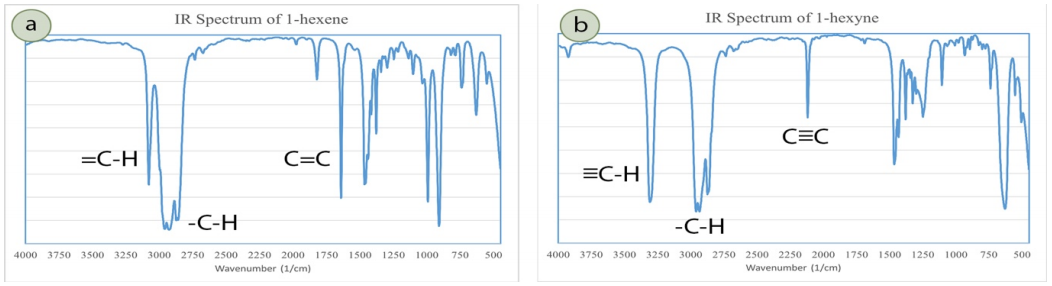
Unsaturated compounds have the following characteristic signals in their IR spectrum:
Alkenes/Aromatics (Figure 6.44a):
- \(\ce{C-H}\) stretch involving \(sp^2\) hybridized carbon atom: medium strength signal slightly above \(3000 \: \text{cm}^{-1}\), normally appearing as a shoulder on the \(\ce{C-H}\) block of signals around \(3000 \: \text{cm}^{-1}\).
- \(\ce{C=C}\) stretch: medium strength signals in the \(1480\)-\(1680 \: \text{cm}^{-1}\) region.
Alkynes (Figure 6.44b):
- \(\ce{C-H}\) stretch involving \(sp\) hybridized carbon atom (terminal alkynes only): strong, sharp signal around \(3300 \: \text{cm}^{-1}\). This signal is in the same region as an \(\ce{O-H}\) signal, but is much sharper.
- \(\ce{C \equiv C}\) triple bond stretch: around \(2100 \: \text{cm}^{-1}\), where few other signals occur. The signal is often weak, and may be absent if the compound is mostly symmetric about the triple bond.
Verification of the presence of an alkene or alkyne can be accomplished with the permanganate and bromine tests. Aromatic groups give no reaction in these tests.
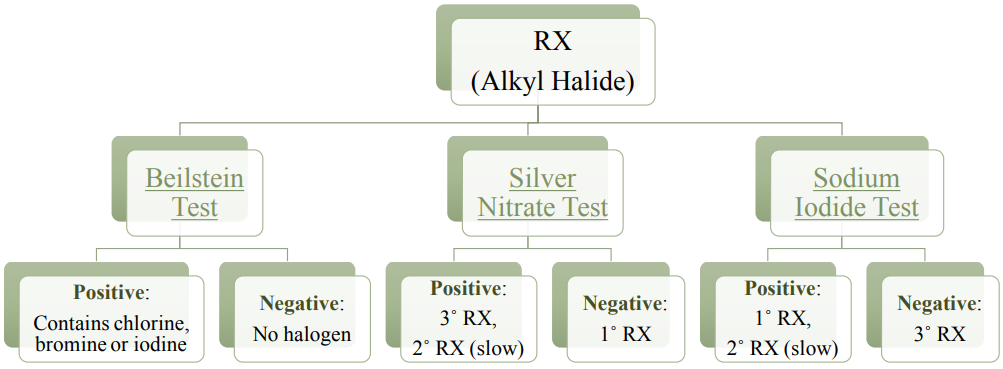
4. Alkyl Halide
Presence of an alkyl halide is not obvious from an IR spectrum, as the \(\ce{C-X}\) signals occur in the "fingerprint region" \(\left( < 1500 \: \text{cm}^{-1} \right)\) and near the lower limit of the IR spectrum (\(\ce{C-Cl}\) stretches occur at \(550\)-\(850 \: \text{cm}^{-1}\) while \(\ce{C-Br}\) stretches occur at \(515\)-\(690 \: \text{cm}^{-1}\)). However, several chemical tests can be used to verify the presence of halogens, the most reliable being the Beilstein test. Two other tests can be used to infer structural features (primary, secondary, tertiary), as shown by the flowchart in Figure 6.45.