5.3B: Fractionating Columns
- Page ID
- 95719
The choice of what fractionating column to use for which application depends in part on availability and the task at hand. Several columns are shown in Figure 5.39: a) Vigreux column with glass indentations, b) Steel wool column made simply be loosely inserting steel wool into the cavity of a fractionating column (similar to a West condenser, but wider), c) Glass beads filled in the cavity of a fractionating column.

These columns have different surface areas and numbers of theoretical plates, and thus differ in their ability to separate close-boiling components. They also differ somewhat in the quantity of compound that will be sequestered through wetting the column. A Vigreux column has the least surface area, making it the least capable of separating close-boiling components. And yet with the lowest surface area, it can have the highest recovery, making it the optimal choice if a separation is not particularly difficult. Glass beads have a high surface area, so are a good choice for separation of close-boiling components. And yet, beads will suffer the greatest loss of material. Steel wool columns have intermediate surface areas and their effectiveness can depend on how tightly the wool is packed in the column. Steel wool columns also cannot be used with corrosive vapors like those containing acid.
To demonstrate the effectiveness of different fractionating columns, a mixture containing \(75 \: \text{mol}\%\) ethylbenzene (normal b.p. \(136^\text{o} \text{C}\)) and \(25 \: \text{mol}\%\) p-cymene (normal b.p. \(177^\text{o} \text{C}\)) was distilled using several methods. The first two \(\text{mL}\) of distillate was collected in each process (Figure 5.40).
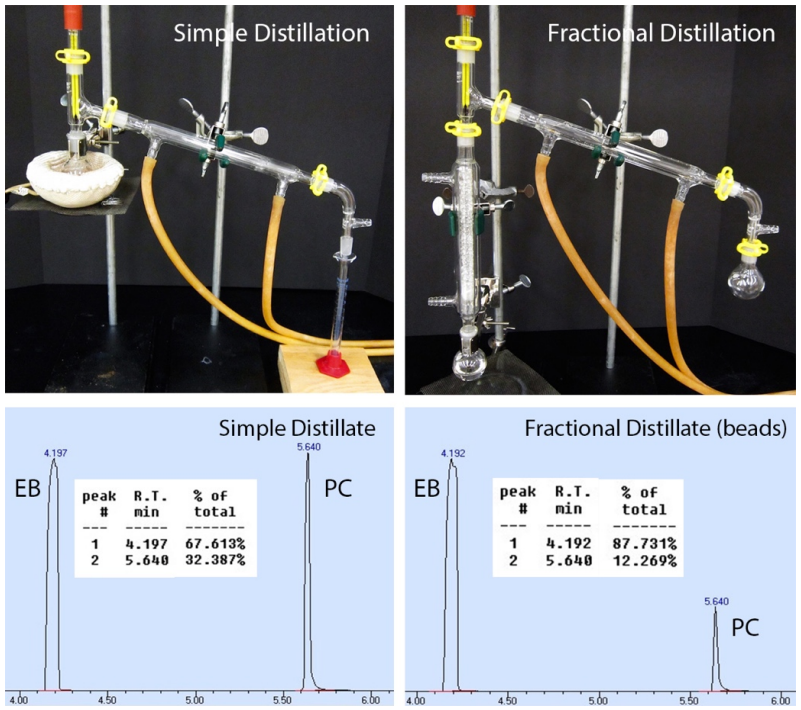
Gas chromatograph analysis of the initial mixture and distillates allowed for quantitation of the mixtures, although a calibration curve was necessary to translate the reported percentages into meaningful values. As the two components had only a \(41^\text{o} \text{C}\) difference in boiling points, a fractional distillation was more effective than a simple distillation, although in some cases only slightly (see Table 5.7). The distillate was never \(> 98\%\) pure with any method as there was a large quantity of the minor component in the distilling pot.
| Method | Components | GC% | Actual % |
|---|---|---|---|
| Initial Solution | ethylbenzene | 55.6% | 75% |
| p-cymene | 44.4% | 25% | |
| Simple Distillation | ethylbenzene | 67.6% | 90% |
| p-cymene | 32.4% | 10% | |
| Fractional (Beads) | ethylbenzene | 87.7% | 97% |
| p-cymene | 12.3% | 3% | |
| Fractional (Steel Wool) | ethylbenzene | 71.4% | 92% |
| p-cymene | 28.6% | 8% | |
| Fractional (Vigreux) | ethylbenzene | 70.4% | 92% |
| p-cymene | 29.6% | 8% |
{{template.HideTOC()}]


