31.6: Vitamin B₁₂ as an Organometallic Compound
- Page ID
- 22408
The structure of vitamin B\(_{12}\) shown in Section 30-6B with a cyanide ion coordinated with cobalt is not the active form of the vitamin but is a particularly stable form, convenient to isolate and handle. The active form is a coenzyme that is remarkable in having a carbon-cobalt bond to an essentially alkyl-type carbon. The carbon-cobalt bond is to a 5'-deoxyadenosyl group, and if we abbreviate vitamin B\(_{12}\) coordinated to cyanide as \(23\), the coenzyme can be written, in the same style, as \(24\). (You will notice that \(23\) is an abbreviation of the formula of Section 30-6B turned \(180^\text{o}\).)
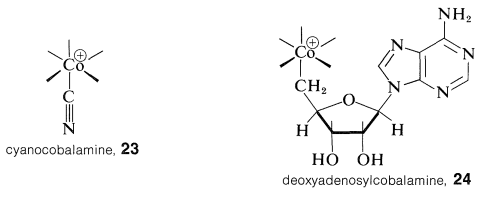
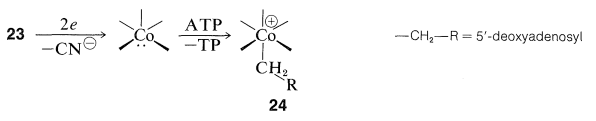
Both \(23\) and the B\(_{12}\) coenzyme, \(24\), are compounds of \(\ce{Co}\)(III) and both substances have all electrons paired. B\(_{12}\) can be reduced to a form with \(\ce{Co}\)(II) which has an unpaired electron and gives an ESR signal (Section 27-9). The cobalt-carbon bond of \(24\) appears to be formed from \(23\) by removal of the cyano group and a two-electron reduction to \(\ce{Co}\)(I). The reduced cobalt is powerfully nucleophilic and probably is alkylated with adenosine triphosphate (ATP, Section 15-5F) to form \(24\):
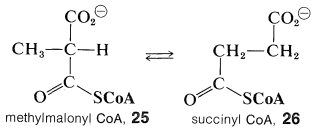
Vitamin B\(_{12}\) coenzyme participates in several biological reactions but none is more unusual, or as hard to rationalize, as its role in the interconversion of methylpropanedioyl CoA (methylmalonyl CoA, \(25\)) to butanedioyl CoA (succinyl CoA, \(26\)):\(^3\)
This rearrangement, which is important in the biochemical utilization of propanoic acid, has been shown to involve transfer of a hydrogen from the \(\ce{CH_3}-\) group of \(25\) to the \(\ce{-CH_2R}\) group of \(24\). Then rearrangement and formation of \(26\) occurs along with reformation of \(24\):
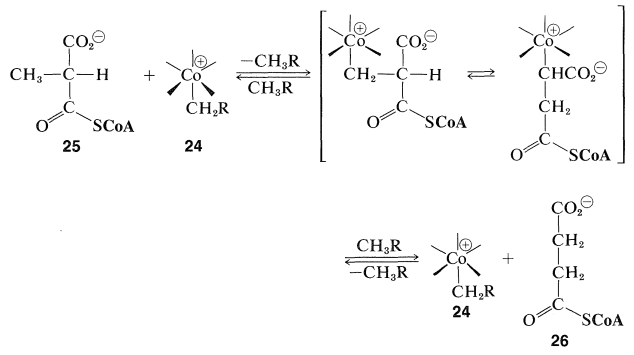
We formulate the intermediate oxidized forms of \(25\) and \(26\) with cobalt-to-carbon bonds, but there is no definitive evidence that this is correct. The overall reaction involves attack on the \(\ce{CH_3}-\) of \(25\), not an easy reaction to carry out in the laboratory, except with reagents such as \(\ce{Cl} \cdot\), because this \(\ce{CH_3}\) is not adjacent to a double bond or other activating group. Furthermore, there is no very good analogy for the rearrangement step. At present, although it is known that \(24\) is reduced to give \(\ce{CH_3-R}\), the details of this important biochemical mechanism remain to be elucidated by further research.
\(^3\)For the structure of CoA, see Section 18-8F.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


