28.8: Chemistry of Vision
- Page ID
- 22382
Vision is a process in which light is absorbed by a pigment in a photoreceptor cell (by a dye in the eye) and the photochemistry that ensues ultimately produces a transient electrical signal that is transmitted to the brain and interpreted as a visual image. There is much that is not fully understood about this process, but we shall discuss briefly the chemistry involved.
The eye is an extraordinarily sensitive instrument. To be sure, its wavelength response is restricted to \(400\)-\(800 \: \text{nm}\), but its degree of sensitivity is such that a fully dark-adapted eye can clearly detect objects in light so dim as to correspond to a light input over the retina of only about 10,000 quanta per second - one light quantum per three minutes per receptor cell in the retina!
The retina is made up of two kinds of light-sensitive (photoreceptor) cells, known as rods and cones. The rods are the more sensitive and are responsible for vision in dim light. The cones are much fewer in number than the rods and provide detail and color vision in good light. The part of the retina that corresponds to the center of the visual field contains only cones. A red pigment called rhodopsin is the photosensitive substance in the rod cells of the retina. It absorbs most strongly in the blue-green region of the visible spectrum \(\left( \lambda_\text{max} \: 500 \: \text{nm} \right)\) and is essentially unaffected by the far-red end of the spectrum. Cone vision appears to involve a different pigment called iodopsin, which absorbs farther toward the red than does rhodopsin.
Rhodopsin is a combination of a protein called opsin, and the highly conjugated aldehyde, 11-cis-retinal:
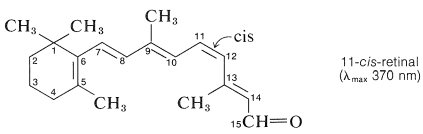
The structure of opsin is unknown, but its prosthetic group (11-cis-retinal) is bonded to it through an imine (Schiff base) function formed between the aldehyde group of the retinal and the side-chain amino function of a lysine unit of opsin:
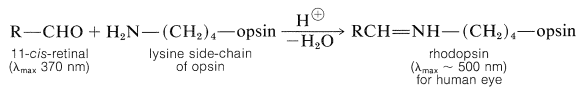
Opsin itself is colorless, whereas 11-cis-retinal absorbs strongly at \(370 \: \text{nm}\). The combination of opsin with 11-cis-retinal produces a remarkable shift of \(\lambda_\text{max}\) to longer wavelengths (\(430 \: \text{nm}\) to \(620 \: \text{nm}\), depending on the species). Similar shifts in wavelength for 11-cis-retinal in combination with simple amines are observed only up to \(\lambda_\text{max}\) of \(440 \: \text{nm}\), and only then for the protonated Schiff base. From this evidence, the chromophore in rhodopsin is believed to be protonated and to be profoundly modified by the structure of the opsin.
Light striking the retina changes the color of rhodopsin from red to yellow. The primary photochemical event in this process was established by G. Wald (Nobel Laureate in Physiology and Medicine, 1967), who showed that light absorption led to a change of configuration about the \(\ce{C_{11}}\)-\(\ce{C_{12}}\) double bond of the retinal chromophore from cis to trans:
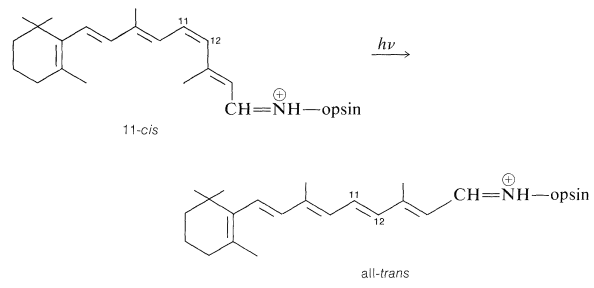
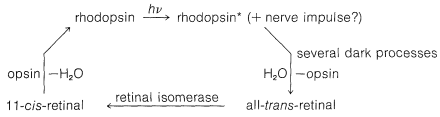
The exact point at which the nerve impulse is transmitted is not established with certainty, but it has to occur before the hydrolysis step because hydrolysis is too slow to account for the nerve impulse. One theory suggests that an electrical signal is generated at the instant of light absorption by electron transfer to a \(\pi \rightarrow \pi^*\) singlet excited state that has substantially charged carbon atoms.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


