22.9: Addition Reactions of Arenes
- Page ID
- 22331
Catalytic Hydrogenation
Benzenoid compounds are not readily converted to cyclohexane derivatives. Nevertheless, several addition reactions are carried out on an industrial scale. Mention was made previously of the hydrogenation of benzene to cyclohexane in the presence of a nickel catalyst:
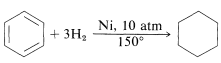
The reaction is very important because cyclohexane is used widely as a solvent and also is oxidized to cyclohexanone, and important intermediate in the synthesis of hexanedioic (adipic) acid and azacycloheptan-2-one (caprolactam), which are used in the preparation of nylon (Section 24-3C).
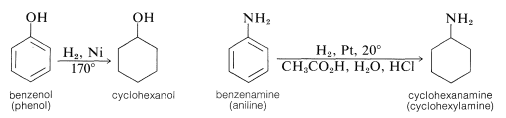
Other cyclohexyl compounds are obtained by catalytic hydrogenation of the corresponding benzene derivatives. Thus cyclohexanol is obtained from benzenol, and cyclohexanamine is obtained from benzenamine (aniline):
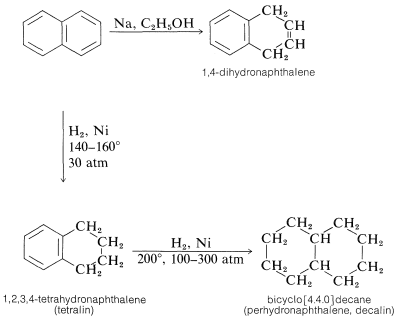
Naphthalene can be reduced more easily than benzene. With sodium in alcohol, 1,4-dihydronaphthalene is formed. Catalytic hydrogenation gives tetralin (1,2,3,4-tetrahydronaphthalene). Further reduction to give perhydronaphthalene (decalin) can be achieved on prolonged catalytic hydrogenation at relatively high temperatures and pressures:
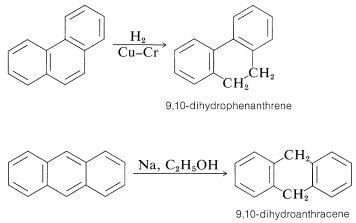
Phenanthrene and anthracene are reduced readily to the dihydro level by addition to the 9,10 positions. Further reduction of the terminal benzene rings is relatively difficult:
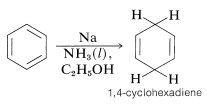
Reduction of Arenes with Metals
Catalytic hydrogenation of benzene cannot be stopped at cyclohexane or cyclohexadiene; it proceeds to cyclohexane. This is because the rate of the first addition step is much slower than of the subsequent steps:
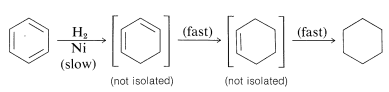
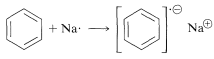
However, benzene and its derivatives can be reduced to cyclohexadienes by solutions of metals such as \(\ce{Li}\), \(\ce{Na}\), \(\ce{K}\), \(\ce{Zn}\), and \(\ce{Hg}\) in a weakly acidic solvent, such as liquid ammonia, amines, or ether-alcohol mixtures. This general type of reaction is known as the Birch reduction after the Australian chemist, A. J. Birch. With benzene, reduction with metals leads to 1,4-cyclohexadiene:
Subsequent steps include a sequence of proton- and electron-transfer steps as follows:
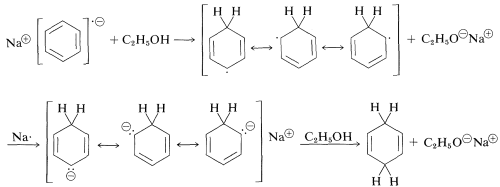
Substituent effects observed for this reaction are entirely consisent with those described for electrophilic substitution and addition - only reversed. That is, the reactivity of an arene in metal reductions is increased by electron-withdrawing groups and decreased by electron-donating groups. Substituents that can stabilize the anion-radical intermediate facilitate the reduction.
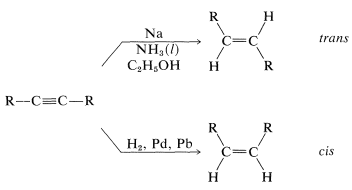
Reduction with metals in weakly acidic solvents is not restricted to arenes. A useful related reaction reduces alkynes to trans-alkenes, and provides a useful alternative to catalytic hydrogenation, which favors formation of cis-alkenes (Section 11-2A):
Halogen Addition
Benzene will add chlorine on irradiation with light to give the fully saturated hexachlorocyclohexane as a mixture of stereoisomers:
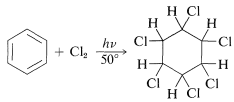
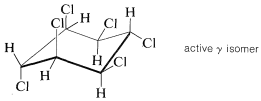
The reaction is commercially important because one of the isomers is a potent insecticide. The product is marketed as a mixture of isomers in which the active isomer \(\left( \gamma \right)\) is optimally about \(40\%\) by weight. It has a variety of trade names: Fortified, BHC, Lindane, Gammexane, Hexachlor.
Cycloaddition
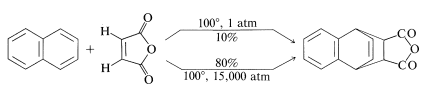
In Chapter 13 we encountered the Diels-Aler reaction, which involves addition of a reactive alkene (dienophile) to the 1,4 positions of a conjugated diene. Neither benzene nor naphthalene reacts significantly with dienophiles on simple heating, but anthracene does react. Cycloaddition occurs between the 9,10 positions:

Reactions in which the transition state has a smaller volume than the reactants are speeded up by an increase in pressure. This is the case with naphthalene and cis-butenedioic anhydride. An \(80\%\) yield of adduct is obtained at \(100^\text{o}\) at 15,000 atmospheres pressure, whereas at one atmosphere and \(100^\text{o}\), the yield is only \(10\%\).
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


