22.6: Orientation in Disubstituted Benzenes
- Page ID
- 22328
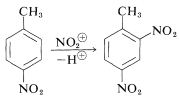
The orientation and reactivity effects of substituents discussed for the substitution of monosubstituted benzenes also hold for disubstituted benzenes, except that the directing influences now come from two groups. Qualitatively, the effects of the two substituents are additive on the reactivity. We therefore would expect 4-nitromethylbenzene to be less reactive than methylbenzene because of the deactivating effect of a nitro group. Also, the most likely position of substiuttion should be, and is, ortho to the methyl group and meta to the nitro group:
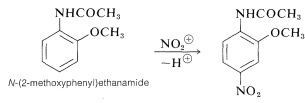
When the two substituents have opposed orientation effects, it is not always easy to predict what products will be obtained. For example, \(\ce{N}\)-(2-methoxyphenyl)ethanamide has two powerful o,p-directing substituents, \(\ce{-OCH_3}\) and \(\ce{-NHCOCH_3}\). Nitration of this compound gives mainly the 4-nitro derivative, which indicates that the \(\ce{-NHCOCH_3}\) exerts a stronger influence than \(\ce{-OCH_3}\):
Seemingly anomalous effects of substituents are known, but such effects may be due to equilibrium control. One example is the aluminum chloride-catalyzed alkylation of benzene, which leads to the formation of a 1,3,5-trialkylbenzene in preference to the expected 1,2,4-isomer (see Section 22-4E). The preferred reaction occurs particularly readily because alkylation is reversible and because alkylation is one of the least selective of the electrophilic aromatic substitutions (considerable meta isomer is formed even under conditions where kinetic control is dominant). Equilibrium control, which favors the 1,3,5-product rather than the less stable 1,2,4-product, becomes most evident when the reaction time, the reaction temperature, and aluminum chloride concentration are increased. Another source of anomalous substituent effects is discussed in the next section.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


