18.7: Functional Derivatives of Carboxylic Acids
- Page ID
- 22292
A functional derivative of a carboxylic acid is a substance formed by replacement of the hydroxyl group of the acid by some other group, \(\ce{X}\), such that it can be hydrolyzed back to the acid in accord with Equation 18-7:
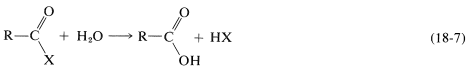
By this definition, an amide, \(\ce{RCONH_2}\), but not a ketone, \(\ce{RCOCH_3}\), is a functional derivative of a carboxylic acid. Several derivatives of carboxylic acids are give in Table 18-3, and methods for preparation of these derivatives are summarized in Tables 18-6 and 18-7 at the end of the chapter.
Table 18-3: Functional Derivatives of Carboxylic Acids
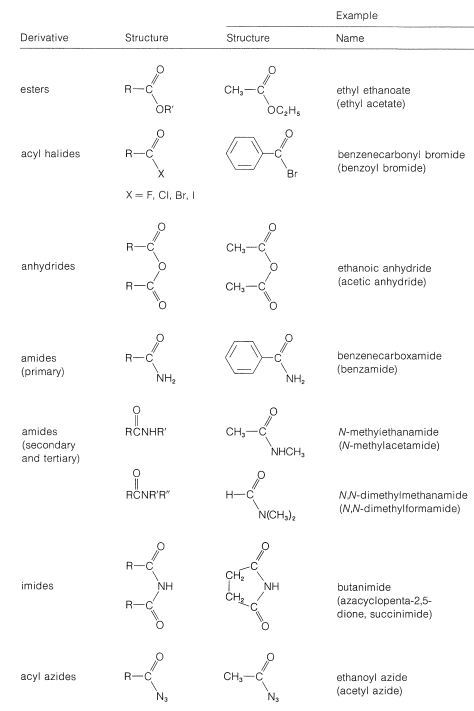
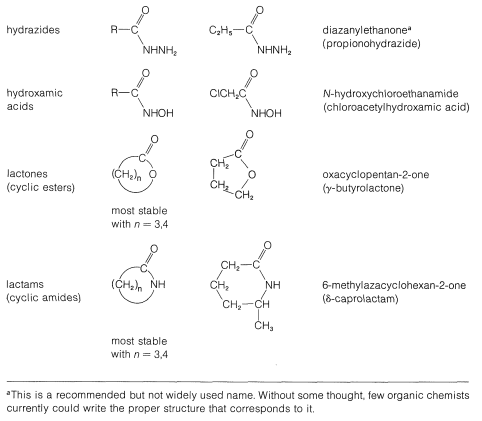
The common structural feature of the compounds listed in Table 18-3 is the acyl group \(\ce{RCO}-\). However, nitriles, \(\ce{RC \equiv N}\), often are considered to be acid derivatives, even though the acyl group is not present as such, because hydrolysis of nitriles leads to carboxylic acids:
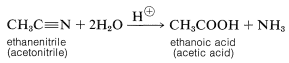
The chemistry of nitriles will be discussed in Section 24-5.
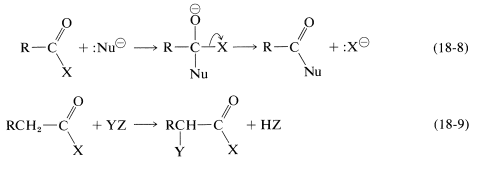
The two main types of reactions of carboxylic acid derivatives with which we now shall be concerned are the replacement of \(\ce{X}\) by attack of a nucleophile \(\ce{Nu}^\ominus\) at the carbonyl carbon with subsequent cleavage of the \(\ce{C-X}\) bond (Equation 18-8), and substitution at the \(\alpha\) carbon facilitated by the carbonyl group (Equation 18-9):
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


