14.8: Polyhalogenated Alkanes and Alkenes
- Page ID
- 21964
Useful Compounds
Polychlorination of methane yields the di-, tri-, and tetrachloromethanes cheaply and efficiently:

These substances have excellent solvent properties for nonpolar and slightly polar substances. Chloroform once was used widely as an inhalation anesthetic. However, it has a deleterious effect on the heart and is oxidized slowly by atmospheric oxygen to highly toxic carbonyl dichloride (phosgene, \(\ce{COCl_2}\)). Commercial chloroform contains about \(1\%\) ethanol, which destroys any \(\ce{COCl_2}\) formed by oxidation.
Carbon tetrachloride commonly was employed as a cleaning solvent, although its considerable toxicity entails considerable hazard when used indiscriminately. It has been used as a fire-extinguishing fluid for petroleum fires, but its toxicity and tendency to form still more toxic carbonyl dichloride makes it undesirable for confined areas. The common laboratory practice of removing traces of water from solvents with metallic sodium should not be applied to halogenated compounds; carbon tetrachloride-sodium mixtures are shock sensitive and can detonate.
Trichloroethene ("Tri-Clene", bp \(87^\text{o}\)) is a widely used dry-cleaning solvent. It can be prepared from either ethene or ethyne:
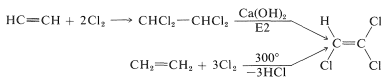
Compared with monohaloalkanes, polyhalogen compounds have quite different reactivities and behavior toward nucleophiles and bases. Thus dichloromethane reacts with hydroxide ion by an \(S_\text{N}2\) mechanism much less readily than methyl chloride. The chloromethanol formed then undergoes a rapid \(E2\) elimination to give methanal (formaldehyde), a substance that exists in water largely as dihydroxymethane:
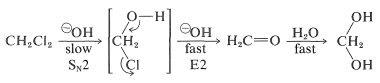
Trichloromethane (chloroform) reacts quite differently with base than does chloromethane or dichloromethane - as will be described in the following section.
\(\alpha\) Elimination. Carbenes
Trihalomethanes, such as trichloromethane (chloroform), are quite reactive toward strong base. The base, such as hydroxide, removes the hydrogen of \(\ce{HCCl_3}\) as a proton much more rapidly than it attacks the carbon in the \(S_\text{N}2\) manner. The carbanion so formed, \(\ce{Cl_3C}^\ominus\), is unstable and loses chloride ion to form a highly reactive neutral intermediate, \(:\ce{CCl_2}\), called dichlorocarbene:
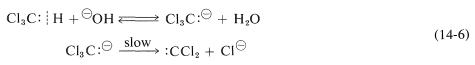
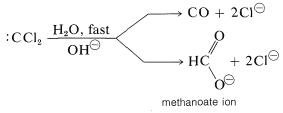
This intermediate has only six valence electrons around carbon and therefore is strongly electrophilic. In aqueous solution it reacts rapidly to form carbon monoxide and methanoate (formate) ion:
The formation of \(:\ce{CCl_2}\) from \(\ce{HCCl_3}\) by the reactions of Equation 14-6 results in the elimination of \(\ce{HCl}\) - the leaving groups, \(\ce{H}\) and \(\ce{Cl}\), both originating from the same carbon atom. Such reactions are not uncommon and are called \(\alpha\) eliminations or 1,1 eliminations to distinguish them from \(E1\) and \(E2\) reactions, which are \(\beta\) eliminations or 1,2 eliminations. Still other possibilities are reactions such as \(\gamma\) or 1,3 eliminations, but these take on the character of internal \(S_\text{N}2\) reactions and will not be considered in detail here.
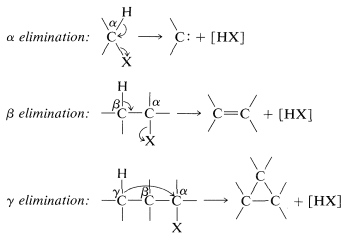
The product of \(\alpha\) elimination is a neutral species that resembles a carbocation in having only six carbon valence electrons. The simplest carbene is \(:\ce{CH_2}\), methylene. Carbenes are highly reactive, so much so that they cannot be isolated. Their involvement in reactions usually has to be inferred from the nature of the products or the reaction kinetics. The characteristic carbene reactions involve forming an electron-pair bond to the carbene carbon by reacting with \(\sigma\) bonds, \(\pi\) bonds, or unshared pairs \(\left( n \right)\). Some of these reactions are illustrated here for methylene \(:\ce{CH_2}\).\(^1\)
with \(\sigma\) bonds (insertion):
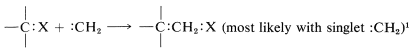
with \(\pi\) bonds ([2 + 1] cycloaddition):
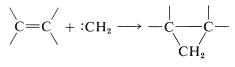
with unshared pairs (dimerization, addition):
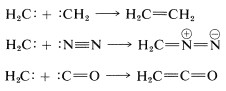
Carbenes are much more reactive toward carbon-carbon double bonds than toward single bonds. Without doubt the most useful feature of \(\alpha\) elimination is that it provides a practical route to cyclopropanes and cyclopropenes by [2 + 1] cycloaddition of carbenes to double or triple bonds. These additions are stereospecific suprafacial additions if they involve singlet carbenes, but can give mixtures with triplet carbenes:
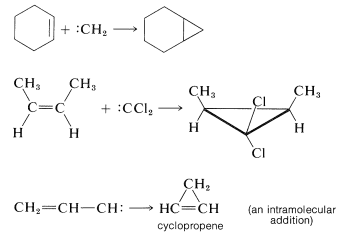
Carbene precursors are compounds that have or acquire good leaving groups (e.g., halide ions). Thus, halogen compounds frequently are carbene sources. Trihalomethanes are the oldest known sources of dihalocarbenes; but there are other methods for generating carbenes, and some of these are listed for reference in Table 14-2 (see also Section 14-10C). There is a question as to whether a “free” carbene actually is formed in some of these reactions, particularly those involving metals, but for our purposes we will classify them as routes to carbenes or carbenelike species.
Table 14-2: \(\alpha\)-Elimination Reactions Producing Carbene Intermediates\(^a\)

Many carbenes, like carbocations, rearrange to more stable structures by the migration of a neighboring group to the electron-deficient carbon. Thus phenylmethylcarbene rearranges to ethenylbenzene (styrene):
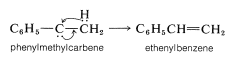
Fluorochloromethanes
Replacement of either one or two of the chlorines of carbon tetrachloride by fluorine can be achieved readily with antimony trifluoride containing some antimony pentachloride. The reaction stops after two chlorines have been replaced. The antimony trifluoride can be regenerated continuously from the antimony chloride by addition of anhydrous hydrogen fluoride:
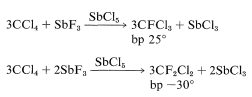
Both products are useful as refrigerants, particularly for household refrigerators and air-conditioning units, under the trade name Freon. Difluorodichloromethane (Freon 12) also is employed as a propellant in aerosol bombs, shaving-cream dispensers, and other such containers. It is nontoxic, odorless, nonflammable, and will not react with hot concentrated mineral acids or metallic sodium. This lack of reactivity is generally characteristic of the difluoromethylene group, provided the fluorines are not located on an unsaturated carbon. Attachment of a fluorine atom to a carbon atom bonded to one or more chlorine atoms tends greatly to reduce the reactivity of the chlorines toward almost all types of reagents. Possible environmental problems associated with these substances were discussed in the introduction to this chapter.
Fluorocarbons
During World War II, plastics and lubricating compounds of unusual chemical and thermal stability were required for many applications, in particular for pumping apparatus used to separate \(\ce{^{235}U}\) from \(\ce{^{238}U}\) by diffusion of corrosive uranium hexafluoride through porous barriers. It was natural to consider the use of substances made only of carbon and fluorine (fluorocarbons) for such purposes, and considerable effort was spent on methods of preparing compounds such as \(\ce{-(CF_2)}-_n\). Today, many such substances are in common use. These often are called "perfluoro-" compounds, which indicates that all available hydrogens of the parent compound are replaced by fluorine. Thus perfluorocyclohexane is \(\ce{(CF_2)_6}\). A widely used perfluorocarbon is the plastic material \(\ce{-(CF_2)}-_n\), which is produced in quantity by radical polymerization of tetrafluoroethene:
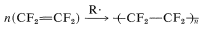
The product ("Teflon") is a solid, chemically inert substance that is stable to around \(300^\text{o}\). It makes excellent electrical insulation and gasket materials. It also has self-lubricating properties, which are exploited in the preparation of low-adhesion surfaces (such as "nonstick" fry pans) and light-duty bearings.
Tetrafluoroethene can be made on a commercial scale by the following method:
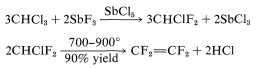
The latter reaction involves difluorocarbene \(\left( :\ce{CF_2} \right)\):
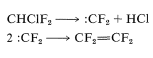
In the presence of peroxides, tetrafluoroethene polymerizes to the long-chain polymer. If peroxides are excluded, [2 + 2] cycloaddition occurs in high yield to give octafluorocyclobutane (see Section 13-3D):
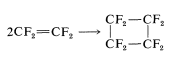
Similar cycloaddition reactions occur with chlorotrifluoroethene and 1,1-dichloro-2,2-difluoroethene.
Radical polymerization of chlorotrifluoroethene gives a useful polymer (Kel-F) that is similar to Teflon.
An excellent elastomer of high chemical resistance (Viton) can be made by copolymerizing hexafluoropropene with 1,1-difluoroethene. The product is stable to \(300^\text{o}\) and is not attacked by hot concentrated nitric acid. Although expensive, it is unrivaled among elastomers for chemical durability under extreme conditions.
Properties of Fluorocarbons
The fluorocarbons have extraordinarily low boiling points relative to the hydrocarbons of comparable molecular weight. As seen in Figure 14-3, their boiling points are nearly the same or even lower than those of the alkanes or cycloalkanes with the same number of carbons. Thus octafluorocyclobutane or boils \(17^\text{o}\) lower than cyclobutane, despite an almost fourfold greater molecular weight!
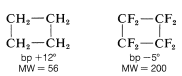
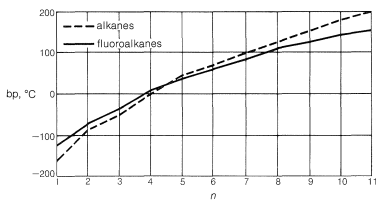
Fluorocarbons are very insoluble in most polar solvents and are only slightly soluble in alkanes in the kerosene range. The higher-molecular-weight fluorocarbons are not even miscible in all proportions with their lower-molecular-weight homologs.
The physiological properties of organofluorine compounds vary widely. Dichlorodifluoromethane and the saturated fluorocarbons appear to be completely nontoxic. In contrast, perfluoro-2-methylpropene is exceedingly toxic, more so than the war gas, carbonyl dichloride \(\left( \ce{COCl_2} \right)\). Sodium fluoroethanoate \(\left( \ce{CH_2FCO_2Na} \right)\) and 2-fluoroethanol are toxic fluorine derivatives of oxygen-containing organic substances. The fluoroethanoate salt is sold commercially as a rodenticide. Interestingly, sodium trifluoroethanoate is nontoxic.
Fluorocarbon derivatives have another interesting and potentially useful property. They dissolve large quantities of oxygen. This fact, combined with their nontoxicity, has led to their use as blood replacements in heart surgery on experimental animals. Mice can live totally immersed in oxygen-saturated liquid fluorocarbons.
\(^1\)Life with carbenes is substantially complicated by the fact that there are two different forms (singlet and triplet) of \(:\ce{CH_2}\) and presumably of all other carbenes. The two forms of \(:\ce{CH_2}\) differ considerably in their reactivity. One is the singlet, which has its unshared electrons paired, while the other is the triplet with the same electrons unpaired. For \(:\ce{CH_2}\), the singlet form is the less stable and more reactive, whereas with \(:\ce{CCl_2}\), the triplet is the less stable and more reactive.
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


