11.4: Hydrogenation with Homogeneous Catalysts
- Page ID
- 22254
Hydrogen addition to multiple bonds is catalyzed by certain complex metal salts in solution. This may be described as homogeneous catalysis and, compared to heterogeneous catalysis, is a relatively new development in the area of hydrogenation reactions. Rhodium and ruthenium salts appear to be generally useful catalysts:
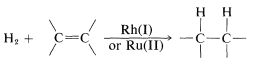
At present, homogeneous catalysis for routine hydrogenation reactions offers little advantage over the convenience and simplicity of heterogeneous catalysis. Suprafacial addition of hydrogen is observed with both types of catalytic systems. However, greater selectivity can be achieved with homogeneous catalysts because they appear to be more sensitive to steric hindrance and are less likely to cause rearrangement, dissociation, and hydrogenation of other bonds (e.g., \(\ce{-NO_2}\) and 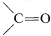 ).
).
The most thoroughly investigated homogeneous hydrogenation catalyst is the four-coordinated rhodium complex \(\ce{Rh} \left[ \ce{(C_6H_5)_3P} \right]_3 \ce{Cl}\). This catalyst is called Wilkinson's catalyst after its discoverer, G. Wilkinson. In 1973, the Nobel Prize in chemistry was awarded jointly to Wilkinson and E. O. H. Fischer for their respective contributions to the field of organometallic chemistry. As you will see in this and later chapters, compounds with carbon-metal bonds (organometallic compounds) are extremely useful reagents, reactive intermediates, or catalysts in organic reactions. To a very large extent, the work of Fischer and Wilkinson created the current interest and developments in the field of transition-metal organic chemistry, which will be discussed in Chapter 31.
Contributors and Attributions
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


