10.6: Electrophilic Addition Reactions of Alkynes
- Page ID
- 22242
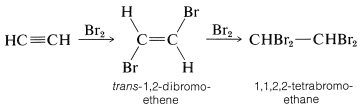
The alkynes behave in many ways as if they were doubly unsaturated alkenes. For example, bromine adds to ethyne in two stages - first to give trans-1,2-dibromoethene by antarafacial addition, and finally to give 1,1,2,2-tetrabromoethane:
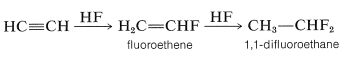
Likewise, anhydrous hydrogen fluoride adds first to give fluoroethene and ultimately to give 1,1-difluoroethane:
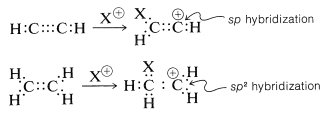
However, there is an interesting contrast in reactivity. Alkynes are substantially less reactive than corresponding alkenes toward many electrophiles. This is perhaps surprising because the electrons of a triple bond, like those of a double bond, are highly exposed, which suggests that the reactivity (nucleophilicity) of a triple bond should be high. Evidently this is not the case. A simple but reasonable explanation is that the carbocation formed from the alkyne is less stable than that from the alkene because it cannot achieve the \(sp^2\) hybrid-orbital configuration expected to be the most stable arrangement for a carbocation (see Section 6-4E):
Hydration of Alkynes
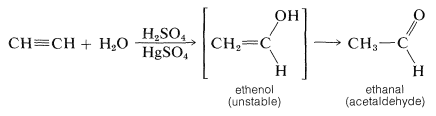
Alkynes, unlike alkenes, are not hydrated readily in aqueous acid unless a mercuric salt is present as a catalyst. Also, the products that are isolated are either aldehydes or ketones instead of alcohols. Even though the addition of one molecule of water to ethyne probably gives ethenol (vinyl alcohol) initially, this compound is unstable relative to its structural isomer (ethanal) and rapidly rearranges:
Similarly, addition of water to propyne leads to 2-propanone by way of its unstable isomer, 2-propenol:
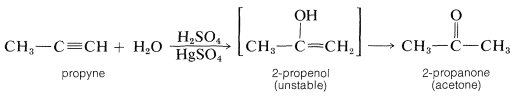
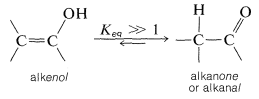
In general, the position of equilibrium for interconversion of a carbonyl compound with the corresponding alkenol (or enol), having the hydroxyl group attached to the double bond, lies far on the side of the carbonyl compound:
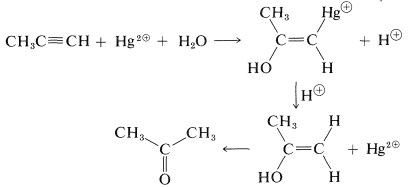
Because mercuric salts catalyze the hydration of alkynes, they probably are acting as electrophiles. Mercuric salts are known to add to both alkenes and alkynes, and if the reaction mixture is acidic, the carbon-mercury bond is cleaved to form a carbon-hydrogen bond. The overall sequence in propyne hydration may be written as follows:
References
John D. Robert and Marjorie C. Caserio (1977) Basic Principles of Organic Chemistry, second edition. W. A. Benjamin, Inc. , Menlo Park, CA. ISBN 0-8053-8329-8. This content is copyrighted under the following conditions, "You are granted permission for individual, educational, research and non-commercial reproduction, distribution, display and performance of this work in any format."


