13.3: Atmospheric Pressure
- Page ID
- 53801

What do storm reports reveal?
The pressure in the atmosphere is an important factor in determining what the weather will be like. If the barometric pressure is high in an area, this will cause air to move to a region of lower pressure. The greater the difference in pressure between the two areas, the stronger the winds will develop. Under certain conditions, the winds can produce a tornado (a violent rotating column of air that reaches from a thunderstorm down to the ground).
Atmospheric Pressure
Atmospheric pressure is the pressure exerted by gas particles in Earth's atmosphere as those particles collide with objects. A barometer is an instrument used to measure atmospheric pressure. A traditional mercury barometer consists of an evacuated tube immersed in a container of mercury. Air molecules push down on the surface of the mercury. Because the inside of the tube is a vacuum, the mercury rises inside the tube. The height to which the mercury rises is dependent on the external air pressure.

A more convenient barometer, called an aneroid barometer, measures pressure by the expansion and contraction of a small spring within an evacuated metal capsule.
Atmospheric Pressure and Altitude
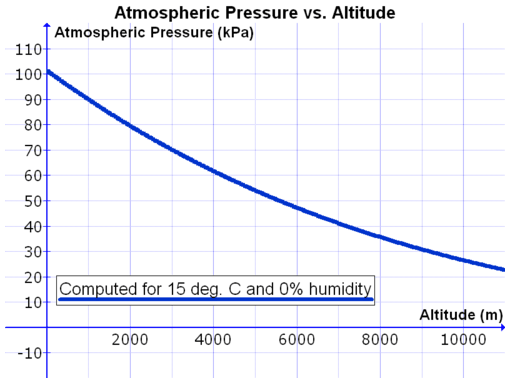
At sea level, a mercury column will rise a distance of \(760 \: \text{mm}\). This atmospheric pressure is reported as \(760 \: \text{mm} \: \ce{Hg}\) (millimeters of mercury). At higher altitudes, the atmospheric pressure is decreased and so the column of mercury will not rise as high. On the summit of Mt. Everest (elevation of \(8848 \: \text{m}\)), the air pressure is \(253 \: \text{mm} \: \ce{Hg}\). Atmospheric pressure is slightly dependent on weather conditions. On the graph below, we can see the decrease in atmospheric pressure as the altitude increases. At sea level, the atmospheric pressure would be a little over \(100 \: \text{kPa}\) (one atmosphere or \(760 \: \text{mm} \: \ce{Hg}\)). If we climb to the top of Mount Everest (the highest mountain in the world at 29,029 feet or 8848 meters), the atmospheric pressure will drop to slightly over \(30 \: \text{kPa}\) (about 0.30 atmospheres or \(228 \: \text{mm} \: \ce{Hg}\)). This marked decrease in atmospheric pressure results in much lower levels of oxygen. Teams that climb this mountain must bring supplies of oxygen with them in order to breathe at these high altitudes.
Ever try cooking at the top of a mountain or at sea level? Compare how atmospheric pressure, vapor pressure and boiling point relate in this simulation:
Summary
- Atmospheric pressure is the pressure exerted by gas particles in Earth’s atmosphere as those particles collide with objects.
- A barometer measures atmospheric pressure.
- Atmospheric pressure decreases as the altitude increases.
Review
- Define atmospheric pressure.
- What is an aneroid barometer?
- How does the atmospheric pressure change as the altitude increases?

