10.3: Water - Both an Acid and a Base
- Page ID
- 16082
- To write chemical equations for water acting as an acid and as a base.
Water (H2O) is an interesting compound in many respects. Here, we will consider its ability to behave as an acid or a base.
In some circumstances, a water molecule will accept a proton and thus act as a Brønsted-Lowry base. We saw an example in the dissolving of HCl in H2O:
\[\rm{HCl + H_2O(ℓ) \rightarrow H_3O^+(aq) + Cl^−(aq)} \label{Eq1} \]
In other circumstances, a water molecule can donate a proton and thus act as a Brønsted-Lowry acid. For example, in the presence of the amide ion (see Example 4 in Section 10.2), a water molecule donates a proton, making ammonia as a product:
\[H_2O(ℓ) + NH^−_{2}(aq) \rightarrow OH^−(aq) + NH_{3}(aq) \label{Eq2} \]
In this case, NH2− is a Brønsted-Lowry base (the proton acceptor).
So, depending on the circumstances, H2O can act as either a Brønsted-Lowry acid or a Brønsted-Lowry base. Water is not the only substance that can react as an acid in some cases or a base in others, but it is certainly the most common example—and the most important one. A substance that can either donate or accept a proton, depending on the circumstances, is called an amphiprotic compound.
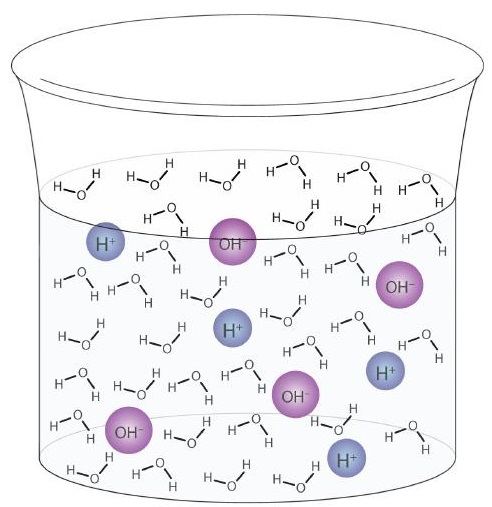
A water molecule can act as an acid or a base even in a sample of pure water. About 6 in every 100 million (6 in 108) water molecules undergo the following reaction:
\[H_2O(ℓ) + H_2O(ℓ) \rightarrow H_3O^+(aq) + OH^−(aq) \label{Eq3} \]
This process is called the autoionization of water (Figure \(\PageIndex{1}\)) and occurs in every sample of water, whether it is pure or part of a solution. Autoionization occurs to some extent in any amphiprotic liquid. (For comparison, liquid ammonia undergoes autoionization as well, but only about 1 molecule in a million billion (1 in 1015) reacts with another ammonia molecule.)
It is rare to truly have pure water. Water exposed to air will usually be slightly acidic because dissolved carbon dioxide gas, or carbonic acid, decreases the pH slightly below 7. Alternatively, dissolved minerals, like calcium carbonate (limestone), can make water slightly basic.
Identify water as either a Brønsted-Lowry acid or a Brønsted-Lowry base.
- H2O(ℓ) + NO2−(aq) → HNO2(aq) + OH−(aq)
- HC2H3O2(aq) + H2O(ℓ) → H3O+(aq) + C2H3O2−(aq)
Solution
- In this reaction, the water molecule donates a proton to the NO2− ion, making OH−(aq). As the proton donor, H2O acts as a Brønsted-Lowry acid.
- In this reaction, the water molecule accepts a proton from HC2H3O2, becoming H3O+(aq). As the proton acceptor, H2O is a Brønsted-Lowry base.
Identify water as either a Brønsted-Lowry acid or a Brønsted-Lowry base.
- HCOOH(aq) + H2O(ℓ) → H3O+(aq) + HCOO−(aq)
- H2O(ℓ) + PO43−(aq) → OH−(aq) + HPO42−(aq)
- Answer
-
1. H2O acts as the proton acceptor (Brønsted-Lowry base)
2. H2O acts as the proton donor (Brønsted-Lowry acid)
Key Takeaway
- Water molecules can act as both an acid and a base, depending on the conditions.


