13.18: Heating and Cooling Curves
- Page ID
- 53816
During the lifetime of Mark Twain (real name: Samuel Langhorne Clemens, 1835-1910), the steamboat was a major means of transportation on the rivers and lakes of the United States. Twain himself was a steamboat pilot on the Mississippi River for a period of time, and took his pen name from the measurement of water depth (twelve feet, which was a safe depth for the boats). The boats got their power from steam—liquid water converted to a gas at high temperatures. The steam would push the pistons of the engine, causing the paddle wheels to turn and propel the boat.
Heating Curves
Imagine that you have a block of ice that is at a temperature of \(-30^\text{o} \text{C}\), well below its melting point. The ice is in a closed container. As heat is steadily added to the ice block, the water molecules will begin to vibrate faster and faster as they absorb kinetic energy. Eventually, when the ice has warmed to \(0^\text{o} \text{C}\), the added energy will start to break apart the hydrogen bonding that keeps the water molecules in place when it is in the solid form. As the ice melts, its temperature does not rise. All of the energy that is being put into the ice goes into the melting process and not into any increase in temperature. During the melting process, the two states—solid and liquid—are in equilibrium with one another. If the system was isolated at that point and no energy was allowed to enter or leave, the ice-water mixture at \(0^\text{o} \text{C}\) would remain. Temperature is always constant during a change of state.
Continued heating of the water after the ice has completely melted will now increase the kinetic energy of the liquid molecules and the temperature will rise. Assuming that the atmospheric pressure is standard, the temperature will rise steadily until it reaches \(100^\text{o} \text{C}\). At this point, the added energy from the heat will cause the liquid to begin to vaporize. As with the previous state change, the temperature will remain at \(100^\text{o} \text{C}\) while the water molecules are going from the liquid to the gas or vapor state. Once all the liquid has completely boiled away, continued heating of the steam (since the container is closed) will increase its temperature above \(100^\text{o} \text{C}\).
The experiment described above can be summarized in a graph called a heating curve (figure below).
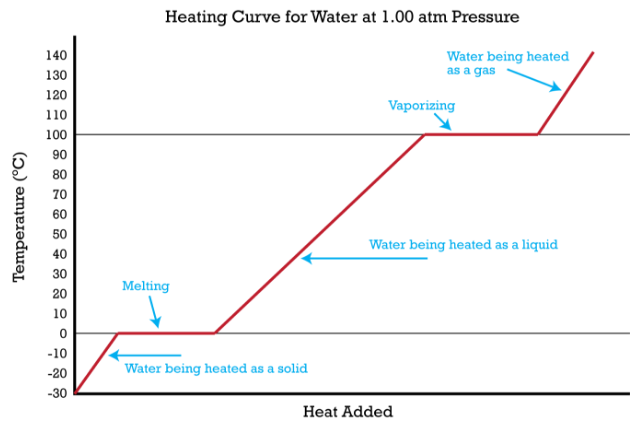
The change of state behavior of all substances can be represented with a heating curve of this type. The melting and boiling points of the substance can be determined by the horizontal lines or plateaus on the curve. Other substances have melting and boiling points that are different from those of water. An exception to this blueprint heating curve is for a substance such as carbon dioxide, which sublimes rather than melts at standard pressure. The heating curve for carbon dioxide would have only one plateau, at the sublimation temperature of \(\ce{CO_2}\).
The entire experiment could be run in reverse. Steam above \(100^\text{o} \text{C}\) could be steadily cooled down to \(100^\text{o} \text{C}\), at which point it would condense to liquid water. The water could then be cooled to \(0^\text{o} \text{C}\), at which point continued cooling would freeze the water to ice. The ice could then be cooled to a point below \(0^\text{o} \text{C}\). This could be diagrammed in a cooling curve that would be the reverse of the heating curve.
Summary of State Changes
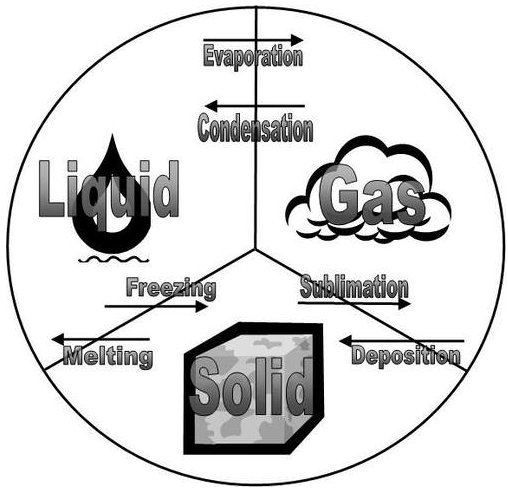
All of the changes of state that occur between solid, liquid, and gas are summarized in the diagram in the figure below. Freezing is the opposite of melting, and both represent the equilibrium between the solid and liquid states. Evaporation occurs when a liquid turns to a gas. Condensation is the opposite of vaporization, and both represent the equilibrium between the liquid and gas states. Deposition is the opposite of sublimation, and both represent the equilibrium between the solid and gas states.
Summary
- A change of state can be brought about by putting heat into a system or removing it from the system.
- The temperature of a system will not change as long as the substance is undergoing a change from solid to liquid to gas, as well as the reverse.
- Freezing is the opposite of melting.
- Evaporation occurs when a liquid turns to a gas.
- Condensation is the opposite of vaporization.
- Deposition is the opposite of sublimation.

