5.E: Stoichiometry and the Mole (Exercises)
- Page ID
- 65011
Exercises (Stoichiometry)
- Think back to the pound cake recipe. What possible conversion factors can you construct relating the components of the recipe?
- Think back to the pancake recipe. What possible conversion factors can you construct relating the components of the recipe?
- What are all the conversion factors that can be constructed from the balanced chemical reaction: \(\ce{2H2(g) + O2(g) → 2H2O(ℓ)}?\)
- What are all the conversion factors that can be constructed from the balanced chemical reaction N2(g) + 3H2(g) → 2NH3(g)?
- Given the chemical equation : Na(s) + H2O(ℓ) → NaOH(aq) + H2(g)
- Balance the equation.
- How many molecules of H2 are produced when 332 atoms of Na react?
- Given the chemical equation: S(s) + O2(g) → SO3(g)
- Balance the equation.
- How many molecules of O2 are needed when 38 atoms of S react?
- For the balanced chemical equation:
6H+(aq) + 2MnO4−(aq) + 5H2O2(ℓ) → 2Mn2+(aq) + 5O2(g) + 8H2O(ℓ)
how many molecules of H2O are produced when 75 molecules of H2O2 react?
-
For the balanced chemical reaction
2C6H6(ℓ) + 15O2(g) → 12CO2(g) + 6H2O(ℓ)how many molecules of CO2 are produced when 56 molecules of C6H6 react?
-
Given the balanced chemical equation
Fe2O3(s) + 3SO3(g) → Fe2(SO4)3how many molecules of Fe2(SO4)3 are produced if 321 atoms of S are reacted?
-
For the balanced chemical equation
CuO(s) + H2S(g) → CuS + H2O(ℓ)how many molecules of CuS are formed if 9,044 atoms of H react?
-
For the balanced chemical equation
Fe2O3(s) + 3SO3(g) → Fe2(SO4)3suppose we need to make 145,000 molecules of Fe2(SO4)3. How many molecules of SO3 do we need?
- One way to make sulfur hexafluoride is to react thioformaldehyde, CH2S, with elemental fluorine:
CH2S + 6F2 → CF4 + 2HF + SF6
If 45,750 molecules of SF6 are needed, how many molecules of F2 are required?
- Construct the three independent conversion factors possible for these two reactions:
- 2H2 + O2 → 2H2O
- H2 + O2 → H2O2
Why are the ratios between H2 and O2 different?
The conversion factors are different because the stoichiometries of the balanced chemical reactions are different.
- Construct the three independent conversion factors possible for these two reactions:
- 2Na + Cl2 → 2NaCl
- 4Na + 2Cl2 → 4NaCl
What similarities, if any, exist in the conversion factors from these two reactions?
Answers
1. \(\dfrac{1\, pound\, butter}{1\, pound\, flour}\) or \(\dfrac{1\, pound\, sugar}{1\, pound\, eggs}\) are two conversion factors that can be constructed from the pound cake recipe. Other conversion factors are also possible.1 pound butter1 pound flour
3. \(\dfrac{2\, molecules\, H_{2}}{1\, molecule\, O_{2}}\) , \(\dfrac{1\, molecule\, O_{2}}{2\, molecules\, H_{2}O}\) , \(\dfrac{2\, molecules\, H_{2}}{2\, molecules\, H_{2}O}\) and their reciprocals are the conversion factors that can be constructed.
5.
- 2Na(s) + 2H2O(ℓ) → 2NaOH(aq) + H2(g)
- 166 molecules
7. 120 molecules
- \(\dfrac{2\, molecules\, H_{2}}{1\, molecule\, O_{2}}\ , \dfrac{1\, molecule\, O_{2}}{2\, molecules\, H_{2}O}\ , \dfrac{2\, molecules\, H_{2}}{2\, molecules\, H_{2}O}\)
- \(\dfrac{1\, molecules\, H_{2}}{1\, molecule\, O_{2}}\ , \dfrac{1\, molecule\, O_{2}}{2\, molecules\, H_{2}O_{2}}\ , \dfrac{1\, molecule\, H_{2}}{1\, molecule\, H_{2}O_{2}}\)
Excercises (The Mole)
- How many atoms are present in 4.55 mol of Fe?
- How many atoms are present in 0.0665 mol of K?
- How many molecules are present in 2.509 mol of H2S?
- How many molecules are present in 0.336 mol of acetylene (C2H2)?
- How many moles are present in 3.55 × 1024 Pb atoms?
- How many moles are present in 2.09 × 1022 Ti atoms?
- How many moles are present in 1.00 × 1023 PF3 molecules?
- How many moles are present in 5.52 × 1025 penicillin molecules?
- Determine the molar mass of each substance.
- Si
- SiH4
- K2O
- Determine the molar mass of each substance.
- Cl2
- SeCl2
- Ca(C2H3O2)2
- Determine the molar mass of each substance.
- Al
- Al2O3
- CoCl3
- Determine the molar mass of each substance.
- O3
- NaI
- C12H22O11
- What is the mass of 4.44 mol of Rb?
- What is the mass of 0.311 mol of Xe?
- What is the mass of 12.34 mol of Al2(SO4)3?
- What is the mass of 0.0656 mol of PbCl2?
- How many moles are present in 45.6 g of CO?
- How many moles are present in 0.00339 g of LiF?
- How many moles are present in 1.223 g of SF6?
- How many moles are present in 48.8 g of BaCO3?
- How many moles are present in 54.8 mL of mercury if the density of mercury is 13.6 g/mL?
- How many moles are present in 56.83 mL of O2 if the density of O2 is 0.00133 g/mL?
Answers
1. 2.74 × 1024 atoms
-
28.086 g
-
32.118 g
-
94.195 g
-
26.981 g
-
101.959 g
-
165.292 g
Exercises (The Mole in Chemical Reactions)
- Express in mole terms what this chemical equation means: CH4 + 2O2 → CO2 + 2H2O
-
Express in mole terms what this chemical equation means.
Na2CO3 + 2HCl → 2NaCl + H2O + CO2 -
How many molecules of each substance are involved in the equation in Exercise 1 if it is interpreted in terms of moles?
-
How many molecules of each substance are involved in the equation in Exercise 2 if it is interpreted in terms of moles?
-
For the chemical equation
2C2H6 + 7O2 → 4CO2 + 6H2Owhat equivalents can you write in terms of moles? Use the ⇔ sign.
-
For the chemical equation
2Al + 3Cl2 → 2AlCl3what equivalents can you write in terms of moles? Use the ⇔ sign.
-
Write the balanced chemical reaction for the combustion of C5H12 (the products are CO2 and H2O) and determine how many moles of H2O are formed when 5.8 mol of O2 are reacted.
-
Write the balanced chemical reaction for the formation of Fe2(SO4)3 from Fe2O3 and SO3 and determine how many moles of Fe2(SO4)3 are formed when 12.7 mol of SO3 are reacted.
-
For the balanced chemical equation
3Cu(s) + 2NO3−(aq) + 8H+(aq) → 3Cu2+(aq) + 4H2O(ℓ) + 2NO(g)how many moles of Cu2+ are formed when 55.7 mol of H+ are reacted?
-
For the balanced chemical equation
Al(s) + 3Ag+(aq) → Al3+(aq) + 3Ag(s)how many moles of Ag are produced when 0.661 mol of Al are reacted?
-
For the balanced chemical reaction
4NH3(g) + 5O2(g) → 4NO(g) + 6H2O(ℓ)how many moles of H2O are produced when 0.669 mol of NH3 react?
-
For the balanced chemical reaction
4NaOH(aq) + 2S(s) + 3O2(g) → 2Na2SO4(aq) + 2H2O(ℓ)how many moles of Na2SO4 are formed when 1.22 mol of O2 react?
-
For the balanced chemical reaction
4KO2(s) + 2CO2(g) → 2K2CO3(s) + 3O2(g)determine the number of moles of both products formed when 6.88 mol of KO2 react.
- For the balanced chemical reaction
Answers
1. One mole of CH4 reacts with 2 mol of O2 to make 1 mol of CO2 and 2 mol of H2O.
Exercises (Mole-Mass and Mass-Mass Calculations)
- What mass of CO2 is produced by the combustion of 1.00 mol of CH4?
CH4(g) + 2O2(g) → CO2(g) + 2H2O(ℓ) -
What mass of H2O is produced by the combustion of 1.00 mol of CH4?
CH4(g) + 2O2(g) → CO2(g) + 2H2O(ℓ) -
What mass of HgO is required to produce 0.692 mol of O2?
2HgO(s) → 2Hg(ℓ) + O2(g) -
What mass of NaHCO3 is needed to produce 2.659 mol of CO2?
2NaHCO3(s) → Na2CO3(s) + H2O(ℓ) + CO2(g) -
How many moles of Al can be produced from 10.87 g of Ag?
Al(NO3) 3(s) + 3Ag → Al + 3AgNO3 -
How many moles of HCl can be produced from 0.226 g of SOCl2?
SOCl2(ℓ) + H2O(ℓ) → SO2(g) + 2HCl(g) -
How many moles of O2 are needed to prepare 1.00 g of Ca(NO3)2?
Ca(s) + N2(g) + 3O2(g) → Ca(NO3) 2(s) -
How many moles of C2H5OH are needed to generate 106.7 g of H2O?
C2H5OH(ℓ) + 3O2(g) → 2CO2(g) + 3H2O(ℓ) -
What mass of O2 can be generated by the decomposition of 100.0 g of NaClO3?
2NaClO3 → 2NaCl(s) + 3O2(g) -
What mass of Li2O is needed to react with 1,060 g of CO2?
Li2O(aq) + CO2(g) → Li2CO3(aq) -
What mass of Fe2O3 must be reacted to generate 324 g of Al2O3?
Fe2O3(s) + 2Al(s) → 2Fe(s) + Al2O3(s) -
What mass of Fe is generated when 100.0 g of Al are reacted?
Fe2O3(s) + 2Al(s) → 2Fe(s) + Al2O3(s) -
What mass of MnO2 is produced when 445 g of H2O are reacted?
H2O(ℓ) + 2MnO4−(aq) + Br−(aq) → BrO3−(aq) + 2MnO2(s) + 2OH−(aq) -
What mass of PbSO4 is produced when 29.6 g of H2SO4 are reacted?
Pb(s) + PbO2(s) + 2H2SO4(aq) → 2PbSO4(s) + 2H2O(ℓ) -
If 83.9 g of ZnO are formed, what mass of Mn2O3 is formed with it?
Zn(s) + 2MnO2(s) → ZnO(s) + Mn2O3(s) -
If 14.7 g of NO2 are reacted, what mass of H2O is reacted with it?
3NO2(g) + H2O(ℓ) → 2HNO3(aq) + NO(g) -
If 88.4 g of CH2S are reacted, what mass of HF is produced?
CH2S + 6F2 → CF4 + 2HF + SF6
- If 100.0 g of Cl2 are needed, what mass of NaOCl must be reacted?
NaOCl + HCl → NaOH + Cl2
Answers
1. 44.0 g
3. 3.00 × 102 g
5. . 0.0336 mol
7. 0.0183 mol
9. 45.1 g
11. 507 g
13. 4.30 × 103 g
15. 163 g
17. 76.7 g
Exercises (Yields)
- What is the difference between the theoretical yield and the actual yield?
-
What is the difference between the actual yield and the percent yield?
-
A worker isolates 2.675 g of SiF4 after reacting 2.339 g of SiO2 with HF. What are the theoretical yield and the actual yield?
SiO2(s) + 4HF(g) → SiF4(g) + 2H2O(ℓ) -
A worker synthesizes aspirin, C9H8O4, according to this chemical equation. If 12.66 g of C7H6O3 are reacted and 12.03 g of aspirin are isolated, what are the theoretical yield and the actual yield?
C7H6O3 + C4H6O3 → C9H8O4 + HC2H3O2 -
A chemist decomposes 1.006 g of NaHCO3 and obtains 0.0334 g of Na2CO3. What are the theoretical yield and the actual yield?
2NaHCO3(s) → Na2CO3(s) + H2O(ℓ) + CO2(g) -
A chemist combusts a 3.009 g sample of C5H12 and obtains 3.774 g of H2O. What are the theoretical yield and the actual yield?
C5H12(ℓ) + 8O2(g) → 5CO2 + 6H2O(ℓ) -
What is the percent yield in Exercise 3?
-
What is the percent yield in Exercise 4?
-
What is the percent yield in Exercise 5?
- What is the percent yield in Exercise 6?
Answers
1. Theoretical yield is what you expect stoichiometrically from a chemical reaction; actual yield is what you actually get from a chemical reaction.
Exercises (Limiting Reagents)
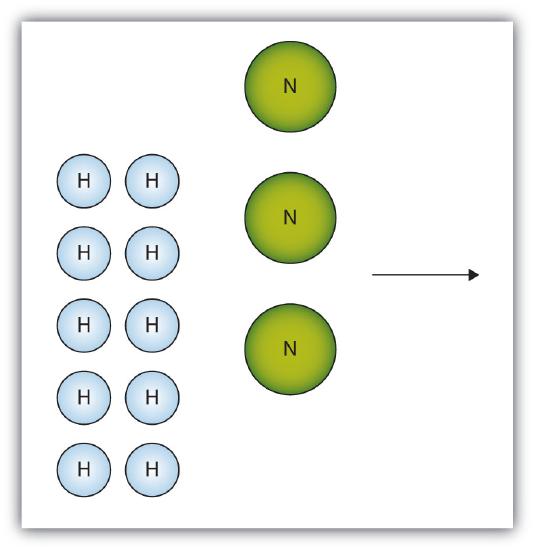
- The box below shows a group of nitrogen and hydrogen molecules that will react to produce ammonia, NH3. What is the limiting reagent?
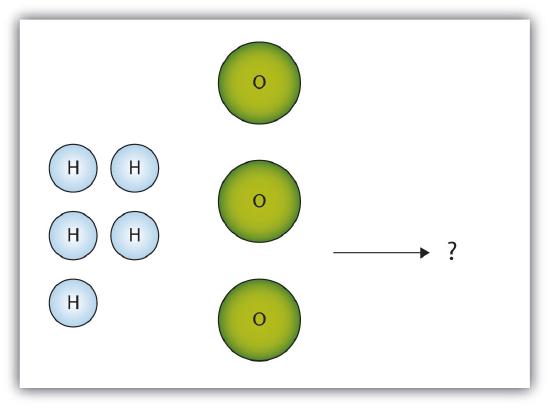
- The box below shows a group of hydrogen and oxygen molecules that will react to produce water, H2O. What is the limiting reagent?
- Given the statement “20.0 g of methane is burned in excess oxygen,” is it obvious which reactant is the limiting reagent?
-
Given the statement “the metal is heated in the presence of excess hydrogen,” is it obvious which substance is the limiting reagent despite not specifying any quantity of reactant?
-
Acetylene (C2H2) is formed by reacting 7.08 g of C and 4.92 g of H2.
2C(s) + H2(g) → C2H2(g)What is the limiting reagent? How much of the other reactant is in excess?
-
Ethane (C2H6) is formed by reacting 7.08 g of C and 4.92 g of H2.
2C(s) + 3H2(g) → C2H6(g)What is the limiting reagent? How much of the other reactant is in excess?
- Given the initial amounts listed, what is the limiting reagent, and how much of the other reactant is in excess?
\[\underset{35.6\, g}{P_{4}O_{6}(s)}+6\underset{4.77\, g}{H_{2}O(l)}\rightarrow 4H_{3}PO_{4}\]
- Given the initial amounts listed, what is the limiting reagent, and how much of the other reactant is in excess?
\[\underset{377\, g}{3NO_{2}(g)}+\underset{244\, g}{H_{2}O(l)}\rightarrow 2HNO_{3}(aq)+NO(g)\]
- To form the precipitate PbCl2, 2.88 g of NaCl and 7.21 g of Pb(NO3)2 are mixed in solution. How much precipitate is formed? How much of which reactant is in excess?
- In a neutralization reaction, 18.06 g of KOH are reacted with 13.43 g of HNO3. What mass of H2O is produced, and what mass of which reactant is in excess?
Answers
1. Nitrogen is the limiting reagent.
Additional Exercises
- How many molecules of O2 will react with 6.022 × 1023 molecules of H2 to make water? The reaction is 2H2(g) + O2(g) → 2H2O(ℓ).
-
How many molecules of H2 will react with 6.022 × 1023 molecules of N2 to make ammonia? The reaction is N2(g) + 3H2(g) → 2NH3(g).
-
How many moles are present in 6.411 kg of CO2? How many molecules is this?
-
How many moles are present in 2.998 mg of SCl4? How many molecules is this?
-
What is the mass in milligrams of 7.22 × 1020 molecules of CO2?
-
What is the mass in kilograms of 3.408 × 1025 molecules of SiS2?
-
What is the mass in grams of 1 molecule of H2O?
-
What is the mass in grams of 1 atom of Al?
-
What is the volume of 3.44 mol of Ga if the density of Ga is 6.08 g/mL?
-
What is the volume of 0.662 mol of He if the density of He is 0.1785 g/L?
-
For the chemical reaction
2C4H10(g) + 13O2(g) → 8CO2(g) + 10H2O(ℓ)assume that 13.4 g of C4H10 reacts completely to products. The density of CO2 is 1.96 g/L. What volume in liters of CO2 is produced?
-
For the chemical reaction
2GaCl3(s) + 3H2(g) → 2Ga(ℓ) + 6HCl(g)if 223 g of GaCl3 reacts completely to products and the density of Ga is 6.08 g/mL, what volume in milliliters of Ga is produced?
-
Calculate the mass of each product when 100.0 g of CuCl react according to the reaction
2CuCl(aq) → CuCl2(aq) + Cu(s)What do you notice about the sum of the masses of the products? What concept is being illustrated here?
-
Calculate the mass of each product when 500.0 g of SnCl2 react according to the reaction
2SnCl2(aq) → SnCl4(aq) + Sn(s)What do you notice about the sum of the masses of the products? What concept is being illustrated here?
-
What mass of CO2 is produced from the combustion of 1 gal of gasoline? The chemical formula of gasoline can be approximated as C8H18. Assume that there are 2,801 g of gasoline per gallon.
-
What mass of H2O is produced from the combustion of 1 gal of gasoline? The chemical formula of gasoline can be approximated as C8H18. Assume that there are 2,801 g of gasoline per gallon.
-
A chemical reaction has a theoretical yield of 19.98 g and a percent yield of 88.40%. What is the actual yield?
-
A chemical reaction has an actual yield of 19.98 g and a percent yield of 88.40%. What is the theoretical yield?
- Given the initial amounts listed, what is the limiting reagent, and how much of the other reactants are in excess?
\[\underset{35.0\, g}{P_{4}}+\underset{12.7\, g}{3NaOH}+\underset{9.33\, g}{3H_{2}O}\rightarrow 2Na_{2}HPO_{4}+PH_{3}\]
- Given the initial amounts listed, what is the limiting reagent, and how much of the other reactants are in excess?
\[\underset{46.3\, g}{2NaCrO_{2}}+\underset{88.2\, g}{3NaBrO_{4}}+\underset{32.5\, g}{2NaOH}\rightarrow 3NaBrO_{3}+2Na_{2}CrO_{4}+H_{2}O\]
- Verify that it does not matter which product you use to predict the limiting reagent by using both products in this combustion reaction to determine the limiting reagent and the amount of the reactant in excess. Initial amounts of each reactant are given.
\[\underset{26.3\, g}{C_{3}H_{8}}+\underset{21.8\, g}{5O_{2}}\rightarrow 3CO_{2}(g)+4H_{2}O(l)\]
- Just in case you suspect Exercise 21 is rigged, do it for another chemical reaction and verify that it does not matter which product you use to predict the limiting reagent by using both products in this combustion reaction to determine the limiting reagent and the amount of the reactant in excess. Initial amounts of each reactant are given.
\[\underset{35.0\, g}{2P_{4}}+\underset{12.7\, g}{6NaOH}+\underset{9.33\, g}{6H_{2}O}\rightarrow 3Na_{2}HPO_{4}+5PH_{3}\]
Answers
1. 1.2044 × 1024 molecules