4.6: Migratory Insertion- Introduction and CO Insertions
- Page ID
- 5076
Introduction
We’ve seen that the metal-ligand bond is generally polarized toward the ligand, making it nucleophilic. When a nucleophilic, X-type ligand is positioned cis to an unsaturated ligand in an organometallic complex, an interesting process that looks a bit like nucleophilic addition can occur.
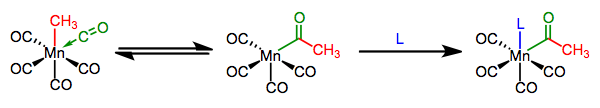
On the whole, the unsaturated ligand appears to insert itself into the M–X bond; hence, the process is called migratory insertion. An open coordination site shows up in the complex, and is typically filled by an added ligand. The open site may appear where the unsaturated ligand was or where the X-type ligand was, depending on which group actually moved (see below). There is no change in oxidation state at the metal (unless the ligand is an alkylidene/alkylidyne), but the total electron count of the complex decreases by two during the actual insertion event—notice in the above example that the complex goes from 18 to 16 total electrons after insertion. A dative ligand comes in to fill that empty coordination site, but stay flexible here: L could be a totally different ligand or a Lewis base in the X-type ligand. L can even be the carbonyl oxygen itself!

We can distinguish between two types of insertions, which differ in the number of atoms in the unsaturated ligand involved in the step. Insertions of CO, carbenes, and other η1 unsaturated ligands are called 1,1-insertions because the X-type ligand moves from its current location on the metal to one spot over, on the atom bound to the metal. η2 ligands like alkenes and alkynes can also participate in migratory insertion; these reactions are called 1,2-insertions because the X-type ligand slides two atoms over, from the metal to the distal atom of the unsaturated ligand.

This is really starting to look like the addition of M and X across a π bond! However, we should take care to distinguish this completely intramolecular process from the attack of a nucleophile or electrophile on a coordinated π system, which is a different beast altogether. Confusingly, chemists often jumble up all of these processes using words like “hydrometalation,” “carbometalation,” “aminometalation,” etc. Another case of big words being used to obscure ignorance! We’ll look at nucleophilic and electrophilic attack on coordinated ligands in separate posts.
Reactivity Trends in CO Insertions
Certain conditions must be met for migratory insertion to occur: the two ligands undergoing the process must be cis, and the complex must be stable with two fewer total electrons. Thermodynamically, the formed Y–X and covalent M–Y bonds must be more stable than the broken M–X and dative M–Y bonds for insertion to be favored. When the opposite is true, the microscopic reverse (elimination or deinsertion) will occur spontaneously.
Migratory aptitudes for insertion into CO have been studied extensively, and the general conclusion here is “it’s complicated.” A few ligands characterized by remarkably stable metal-ligand bonds don’t undergo insertion for thermodynamic reasons—the M–X bond is just too darn strong. Perfluoroalkyl complexes and metal hydrides are two notable examples. Electron-withdrawing groups on the X-type ligand, which strengthen the M–X bond, slow down insertion (likely for thermodynamic reasons though…Hammond’s postulate in action).
What factors affect the relative speed (kinetics) of favorable insertions? Sterics is one important variable. Both 1,1- and 1,2-insertions can relieve steric strain at the metal center by spreading out the ligands involved in the step. In 1,2-insertions, the X-type ligand removes itself completely from the metal! Unsurprisingly, then, bulky ligands undergo insertions more rapidly than smaller ligands. Complexes of the first-row metals tend to react more rapidly than analogous second-row metal complexes, and second-row metal complexes react faster than third-row metal complexes. This trend fits in nicely with the typical trend in M–C bond strengths: first row < second row < third row. Lewis acids help accelerate insertions into CO by coordinating to CO and making the carbonyl carbon more electrophilic. For a similar reason, CO ligands bound to electron-poor metal centers undergo insertion more rapidly than CO’s bound to electron-rich metals. Finally, for reasons that are still unclear, one-electron oxidation often increases the rate of CO insertion substantially.
Although the thermodynamics of alkene 1,2-insertion are more favorable for metal-carbon than metal-hydrogen bonds, M–H bonds react much more rapidly than M–C bonds in 1,2-insertions. This fact has been exploited for olefin hydrogenation, which would be much less useful if it had to complete with olefin polymerization (the result of repeated insertion of C=C into M–C) in the same reaction flask! More on that in the next post.
Stereochemistry in CO Insertions
Migratory insertion steps are full of stereochemistry! Configuration at the migrating alkyl group is retainedduring insertion—a nice piece of evidence supporting a concerted, intramolecular mechanism of migration.
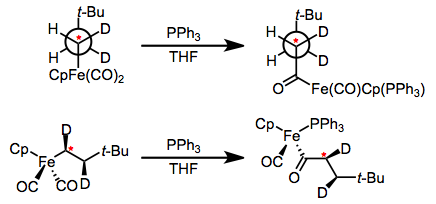
What about stereochemistry at the metal center? Migratory insertion may create a stereogenic center at the metal—see the iron example above. Whether the X-type ligand moves onto the unsaturated ligand or vice versa will impact the configuration of the product complex. Calderazzo’s study of this issue is one of my favorite experiments in all of organometallic chemistry! He took the simple labeled substrate in the figure below and treated it with dative ligand, encouraging insertion. Four products of insertion are possible, corresponding to reaction of the four CO ligands cis to the methyl ligand. Try drawing a few curved arrows to wrap your mind around the four possibilities, and consider both CO migration and Me migration as possible at this point.

Note that product D is impossible if we only allow the Me group to migrate—the spot trans to the labeled CO is another CO ligand, so that spot can only pick up L if CO migrates (not if Me migrates). On the other hand, product C must have come from the migration of Me, since the Me group has moved from a cis to a transposition relative to the labeled CO in product C. Calderazzo observed products A, B, and C, but not D, supporting a mechanism involving Me migration. Other experiments since support the idea that most of the time, the alkyl group migrates onto CO. Slick, huh?
I won’t address insertions into alkylidenes, alkylidynes, and other one-atom unsaturated ligands in this post, as insertions into CO are by far the most popular 1,1 insertions in organometallic chemistry. In the next post, we’ll dig more deeply into 1,2-insertions of alkenes and alkynes. Thanks for reading!


