Geometry of Complex Ions
- Page ID
- 3706
This page describes the shapes of some common complex metal ions. These shapes are for complex ions formed using monodentate ligands - ligands which only form one bond to the central metal ion. You will probably be familiar with working out the shapes of simple compounds using the electron pair repulsion theory. Unfortunately that does not work for most complex metal ions involving transition metals.
6-coordinated Complex Ions
These are complex ions in which the central metal ion is forming six bonds. In the simple cases we are talking about, that means that it will be attached to six ligands. These ions have an octahedral shape. Four of the ligands are in one plane, with the fifth one above the plane, and the sixth one below the plane. The diagram shows four fairly random examples of octahedral ions.
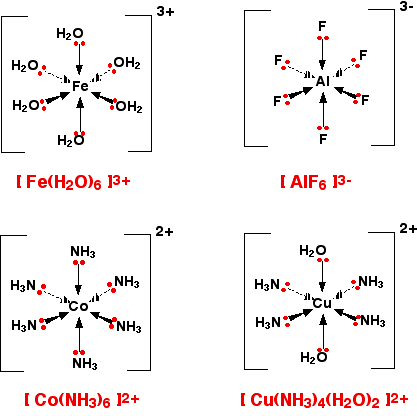
It does not matter the nature of the ligands. If you have six of them, this is the shape they will take up. Easy!
4-coordinated Complex Ions
These are far less common, and they can take up one of two different shapes.
Tetrahedral ions
There are two very similar ions which crop up commonly at this level: [CuCl4]2- and [CoCl4]2-. The copper(II) and cobalt(II) ions have four chloride ions bonded to them rather than six, because the chloride ions are too big to fit any more around the central metal ion.
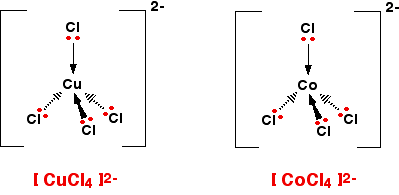
A square planar complex
Occasionally a 4-coordinated complex turns out to be square planar. There is no easy way of predicting that this is going to happen. The only one you might possibly come across at this level is cisplatin which is used as an anti-cancer drug. Cisplatin is a neutral complex, Pt(NH3)2Cl2. It is neutral because the 2+ charge of the original platinum(II) ion is exactly canceled by the two negative charges supplied by the chloride ions.
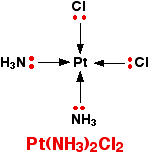
The platinum, the two chlorines, and the two nitrogens are all in the same plane. We will have more to say about cisplatin immediately below.
Contributors and Attributions
Jim Clark (Chemguide.co.uk)


