20.9A: The Nephelauxetic Effect
- Page ID
- 34406
The spectra of the aqua ions for some first row transition metal ions are shown below.

For a much more detailed description of the interpretation of the spectra of first row transition metal ion complexes see the notes on the use of Tanabe-Sugano diagrams.
Cr(III) - an example in more detail
For \9d^3\), \(d^8\) octahedral and \(d^2\), \(d^7\) tetrahedral complexes, the above diagrams can be used to interpret the observed electronic absorption spectra.
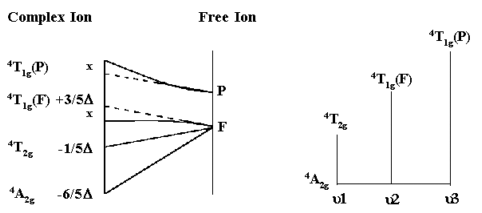
Take for example the \(\ce{Cr^{3+}}\) aquo-ion \(\ce{ [Cr(H2O)6]^{3+}}\). From the simplified Orgel diagram in Figure 2, three absorptions transitions are expected. In practice, the spectrum is found to contain three bands which occur at 17,000 cm-1, 24,000 cm-1 and 37,000 cm-1. Of which only two are shown in Figure 1.
\(μ_1\) corresponds exactly to Δ (Delta) and since the lowest band is found at 17,000 cm-1 then this enables us to measure Δ directly from the spectrum.
The next band is found at 24,000 cm-1 and this can be equated to:
\[μ_2=9/5Δ - x\]
where \(x\) is the configuration interaction between the T(F) state and the T(P) state of the same symmetry.
Since \(Δ\) is 17,000 and μ2 is observed at 24,000 then x must be 6,600 cm-1.
The last band is seen at 37,000 cm-1 and here
\[μ_3=6/5Δ + 15B + x \label{eq3}\]
where \(B\) is one of the RACAH parameters.
Solving Equation \ref{eq3} for \(B\) gives a value of \(B=667\,cm^{-1}\).
For the free Cr3+ ion, B is ~1030 cm-1 so that in the complex this term is reduced by ~2/3 of the free ion value.
A large reduction in B indicates a strong Nephelauxetic Effect. The Nephelauxetic Series is given by:
\[\ce{F^{-} > H2O > urea > NH3 > en \~ C2O42^{-} > NCS^{-} > Cl^{-} ~ CN^{-} > Br^{-} > S2^{-} ~ I^-}\]
Ionic ligands such as \(\ce{F^{-}}\) give a small reduction in \(B\), while covalently bonded ligands such as \(\ce{I^{-}}\) give a large reduction of \(B\).
Contributors and Attributions
Prof. Robert J. Lancashire (The Department of Chemistry, University of the West Indies)


