8.1: Metal Carbonyls
- Page ID
- 172766
Learning Objectives
In this lecture you will learn the following
- The historical background of metal carbonyl complexes.
- The CO ligand and its binding ability to metal.
- Synergism between the ligand to metal forward σ–donation and the metal to ligand backward π–donation observed in a metal-CO interaction.
- The synthesis, characterization and their reactivity of the metal carbonyl compounds.
Metal carbonyls are important class of organometallic compounds that have been studied for a long time. Way back in 1884, Ludwig Mond, upon observing that the nickel valves were being eating away by CO gas in a nickel refining industry, heated nickel powder in a stream of CO gas to synthesize the first known metal carbonyl compound in the form Ni(CO)4. The famous Mond refining process was thus born, grounded on the premise that the volatile Ni(CO)4 compound can be decomposed to pure metal at elevated temperature. Mond subsequently founded the Mond Nickel Company Limited for purifying nickel from its ore using this method.
The carbonyl ligand (CO) distinguishes itself from other ligands in many respects. For example, unlike the alkyl ligands, the carbonyl (CO) ligand is unsaturated thus allowing not only the ligand to σ−donate but also to accept electrons in its π* orbital from dπ metal orbitals and thereby making the CO ligand π−acidic. The other difference lies in the fact that CO is a soft ligand compared to the other common σ−and π−basic ligands like H2O or the alkoxides (RO−), which are considered as hard ligands.
Being π−acidic in nature, CO is a strong field ligand that achieves greater d−orbital splitting through the metal to ligand π−back donation. A metal−CO bonding interaction thus comprises of a CO to metal σ−donation and a metal to CO π−back donation (Figure \(\PageIndex{1}\)). Interestingly enough, both the spectroscopic measurements and the theoretical studies suggest that the extent of the metal to CO π−back donation is almost equal to or even greater than the extent of the CO to metal σ−donation in metal carbonyl complexes. This observation is in agreement with the fact that low valent−transition metal centers tend to form metal carbonyl complexes.
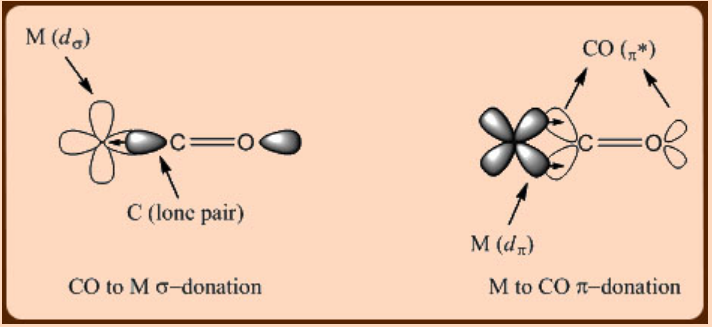
In the metal carbonyl complexes, the direct bearing of the π−back donation is observed on the M−C bond distance that becomes shorter as compared to that of a normal M−C single bond distance. For example, the CpMo(CO)3CH3 complex, exhibits two kind of M−C bond distances that comprise of a longer Mo−CH3 distance (2.38 Å) and a much shorter Mo−CO distance (1.99 Å) arising out of a metal to ligand π−back donation. It becomes thus apparent that the metal−CO interaction can be easily characterized using X−ray crystallography. The infrared spectroscopy can also be equally successfully employed in studying the metal−CO interaction. Since the metal to CO π−back bonding involves a π−donation from the metal dπ orbital to a π* orbital of a C−O bond, significant shift of the ν(CO) stretching frequency towards the lower energy is observed in metal carbonyl complexes with respect to that of free CO (2143 cm−1).
Preparation of metal carbonyl complexes
The common methods of the preparation of the metal carbonyl compounds are,
i. Directly using CO
\[\ce{Fe ->[CO, \: 200atm, \: 200°C] Fe(CO)5} \nonumber \]
The main requirement of this method is that the metal center must be in a reduced low oxidation state in order to facilitate CO binding to the metal center through metal to ligand π−back donation.
ii. Using CO and a reducing agent
\[\ce{NiSO4 + CO + S2O4^{2-} -> Ni(CO)4} \nonumber \]
This method is commonly called reductive carbonylation and is mainly used for the compounds having higher oxidation state metal centers. The reducing agent first reduces the metal center to a lower oxidation state prior to the binding of CO to form the metal carbonyl compounds.
iii. From carbonyl compounds
This method involves abstraction of CO from organic compounds like the alcohols, aldehydes and CO2.
Reactivities of metal carbonyls
i. Nucleophilic attack on carbon
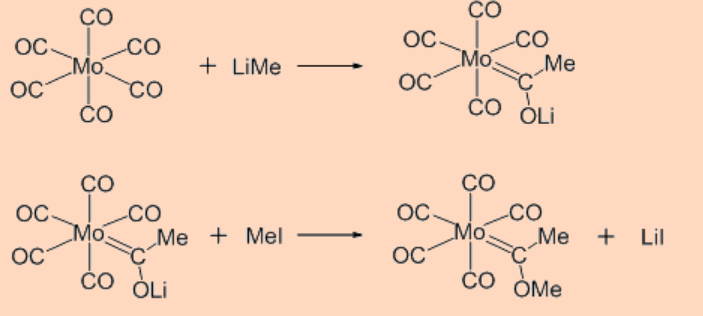
The reaction usually gives rise to carbene moiety.
ii. Electrophilic attack at oxygen
\[\ce{Cl(PR3)4Re-CO + AlMe3 -> Cl(PR3)4Re-CO -> AlMe3} \nonumber \]
iii. Migratory insertion reaction
\[\ce{MeMn(CO)5 +PMe3 -> (MeCO)Mn(CO)4(PMe3)} \nonumber \]
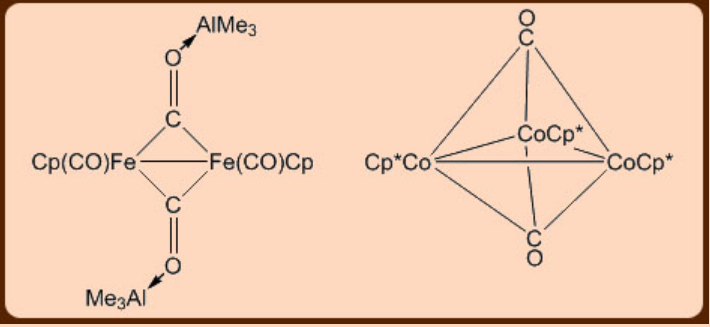
The metal carbonyl displays two kinds of bindings in the form of the terminal and the bridging modes. The infrared spectroscopy can easily distinguish between these two binding modes of the metal carbonyl moiety as the terminal ones show ν(CO) stretching band at ca. 2100-2000 cm−1 while the bridging ones appear in the range 1720−1850 cm−1. The carbonyl moiety can bridge between more than two metal centers (Figure \(\PageIndex{2}\)).
Problems
1. How many lone pairs are there in the CO molecule?
Ans: Three (one from carbon and two from oxygen).
2. Despite O being more electronegative than C, the dipole moment of CO is almost zero. Explain.
Ans: Because of the electron donation from oxygen to carbon.
3. What type of metal centers form metal carbonyl complexes?
Ans: Low−valent metal centers.
4. What are the two main modes of binding exhibited by CO ligand?
Ans: Terminal and bridging modes of binding.
Self Assessment test
1. Predict the product of the reaction?
\[\ce{Ni + CO_{Excess} ->} \nonumber \]
Ans: Three (one from carbon and two from oxygen).
2. Upon binding to a metal center the C−O stretching frequency increases/decreases with regard to that of the free CO?
Ans: Decreases.
3. Explain why do low−valent metal centers stabilize CO binding in metal carbonyl complexes?
Ans: Because metal to ligand π−back donation.
4. Give an example of a good σ−donor and π−donor ligand?
Ans: Alkoxides (RO-).
Summary
CO is a hallmark ligand of organometallic chemistry. The metal carbonyl complexes have been studied for a long time. The CO ligands bind tightly to metal center using a synergistic mechanism that involves σ−donation of the ligand lone pair to metal and followed by the π−back donation from a filled metal d orbital to a vacant σ* orbital of C−O bond of the CO ligand. The metal carbonyl complexes are prepared by several methods. The metal carbonyl complexes are usually stabilized by metal centers in low oxidation states.


