7.5: Amorphous Alloys
- Page ID
- 183336
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \) \( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)\(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\) \(\newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\) \( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\) \( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\) \( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\) \( \newcommand{\Span}{\mathrm{span}}\)\(\newcommand{\AA}{\unicode[.8,0]{x212B}}\)

Alloys of metals with more complex stoichiometries can be made in amorphous form by slower cooling from the melt. These alloys have been prepared and studied since the 1960s, and since the 1990s amorphous alloys have been discovered that can be prepared in bulk form at cooling rates on the order of 1 deg/s, similar to the cooling rates of other kinds of glasses.
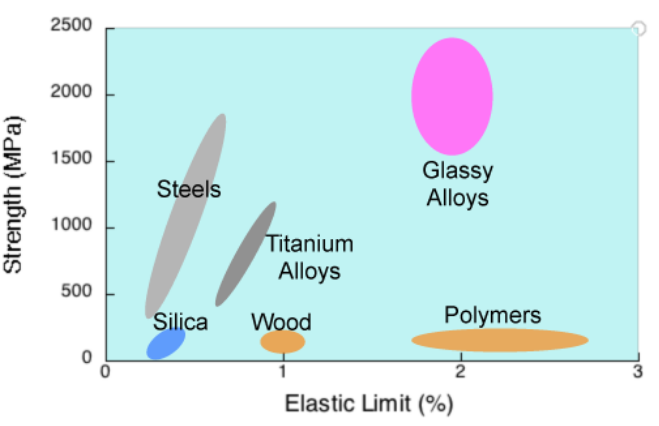
Currently amorphous metals (marketed under the tradenames Vitreloy and Liquidmetal) are used commercially in golf clubs, watches, USB flash drives, and other applications where very high elasticity, yield strength, and/or wear resistance are needed.
| Year | Alloy | Cooling Rate (K/s) |
|---|---|---|
| 1960 | Au75Si24 | 106 - thin films & ribbons[3] |
| 1969 | Pd-Cu-Si | 100-1000 |
| 1980s | La-Al-Cu & others | 1-100 |
| 1990s | Zr-Ti-Cu-Ni-Be | ~1 (similar to oxide glasses) |


