2.8: Three-center Bonding
- Page ID
- 183906
Many (but not all) of the problems we will solve with MO theory derive from the MO diagram of the H2 molecule (Fig. 2.1.5), which is a case of two-center bonding. The rest we will solve by analogy to the H3+ ion, which introduces the concept of three-center bonding.
We can draw the H3+ ion (and also H3 and H3-) in either a linear or triangular geometry.
Walsh correlation diagram for H3+:
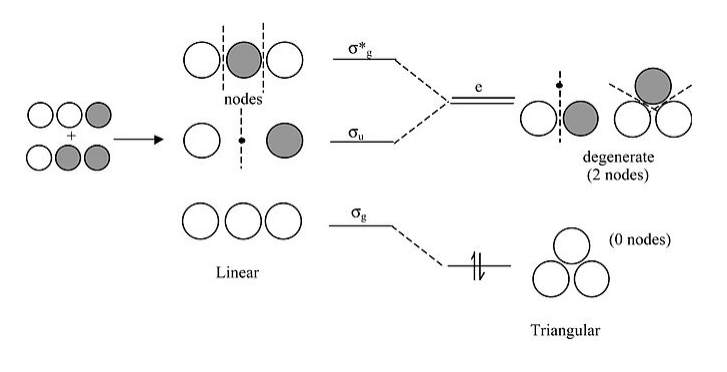
A few important points about this diagram:
- For the linear form of the ion, the highest and lowest MO’s are symmetric with respect to the inversion center in the molecule. Note that the central 1s orbital has g symmetry, so by symmetry it has zero overlap with the u combination of the two 1s orbitals on the ends. This makes the σu orbital a nonbonding orbital.
- In the triangular form of the molecule, the orbitals that derive from σu and σ*g become degenerate (i.e., they have identically the same energy by symmetry). The term symbol “e” means doubly degenerate. We will see later that “t” means triply degenerate. Note that we drop the “g” and “u” for the triangular orbitals because a triangle does not have an inversion center.
- The triangular form is most stable because the two electrons in H3+ have lower energy in the lowest orbital. Bending the molecule creates a third bonding interaction between the 1s orbitals on the ends.
MO diagram for XH2 (X = Be, B, C…):
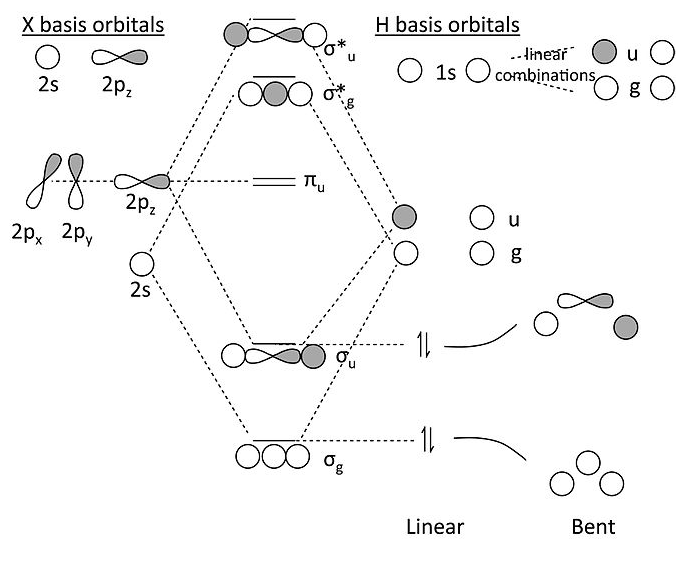
Some key points about this MO diagram:
- In the linear form of the molecule, which has inversion symmetry, the 2s and 2p orbitals of the X atom factor into three symmetry classes:
- 2s = σg
- 2pz = σu
- 2px, 2py = πu
- Similarly, we can see that the two H 1s orbitals make two linear combinations, one with σg symmetry and one with σu symmetry. They look like the bonding and antibonding MO’s of the H2 molecule (which is why we say we use that problem to solve this one).
- The πu orbitals must be non-bonding because there is no combination of the H 1s orbitals that has πu symmetry.
- In the MO diagram, we make bonding and antibonding combinations of the σg’s and the σu’s. For BeH2, we then populate the lowest two orbitals with the four valence electrons and discover (not surprisingly) that the molecule has two bonds and can be written H-Be-H. The correlation diagram shows that a bent form of the molecule should be less stable.
An interesting story about this MO diagram is that it is difficult to predict a priori whether CH2 should be linear or bent. In 1970, Charles Bender and Henry Schaefer, using quantum chemical calculations, predicted that the ground state should be a bent triplet with an H-C-H angle of 135°.[4] The best experiments at the time suggested that methylene was a linear singlet, and the theorists argued that the experimental result was wrong. Later experiments proved them right!
“A theory is something nobody believes, except the person who made it. An experiment is something everybody believes, except the person who made it.” – Einstein


