7.7: Suboxides of Carbon
- Page ID
- 212896
Carbon suboxide
Carbon suboxide is the third oxide of carbon, C3O2. It is made from the dehydration of malonic acid, (7.7.1), with P4O10 above 140 °C Like carbon dioxide, the C3O2 molecule is linear, with pπ-pπ bonding.
\[ \text{CH}_2\text{(CO}_2\text{H)}_2 \rightarrow \text{O=C=C=C=O + 2 H}_2\text{O}\]
Gaseous carbon suboxide has an evil smell and while stable at –78 °C it polymerizes at 25 °C. Photolysis of C3O2 yields the unstable C2O. As expected from its synthesis, carbon suboxide reacts slowly with water to form malonic acid, i.e., the reverse of (7.7.1); however, the reaction with stronger nuceophiles such as amines is rapid, (7.7.2).
\[ \text{C}_3\text{O}_2\text{ + 2 HNR}_2 \rightarrow \text{R}_2\text{NC(O)CH}_2\text{C(O)NR}_2\]
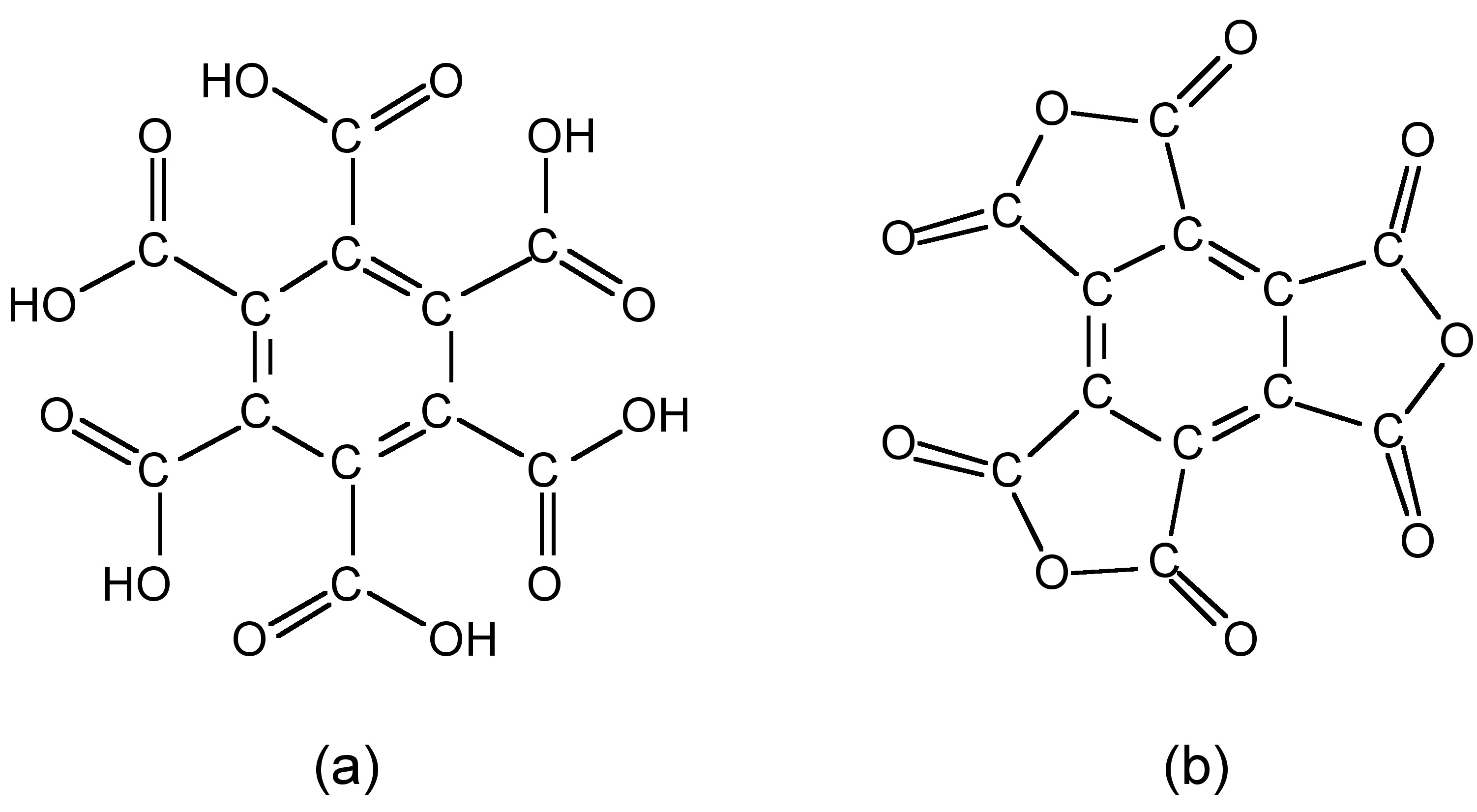
Mellitic acid anhydride
The anhydride of mellitic acid (Figure \(\PageIndex{1}\)a) may be considered as an oxide of carbon since its chemical formula contains only carbon and oxygen, i.e., C12O9 (Figure \(\PageIndex{1}\)b).