4.3: Differences for Beryllium and Magnesium
- Page ID
- 212632
While the chemistry of strontium, barium (and radium) is similar to that of calcium, magnesium and beryllium show marked differences. In both cases these differences are due to the small size of the ions.
Beryllium
Beryllium can be thought of as being even more covalent than magnesium. The small size (ca. 0.3 Å) results in a very high charge density of Be2+. In addition, the ionization energy for beryllium is a large positive value (1st ionization energy = 899.5 kJ/mol, 2nd ionization energy = 14,848.7 kJ/mol). Both of these factors means that the free ion does not exist. Instead, beryllium forms covalent compounds in a similar manner to its diagonal analog aluminum. Both beryllium and aluminum form covalent compounds or strongly solvated cations, and both form polymeric hydrides, chlorides, and alkyls.
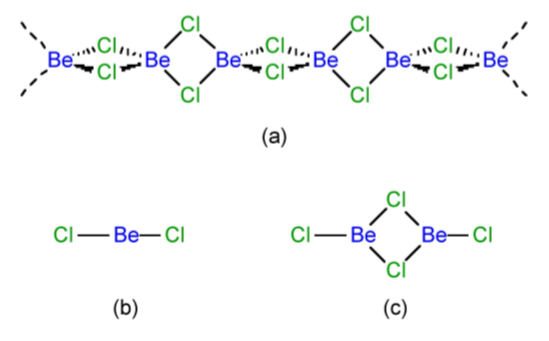
Beryllium chloride is not a lattice structure with a concomitantly high melting and boiling point as observed for the other Group 2 metals (Table4.8). Instead BeCl2 is a polymer in the solid state (Figure \(\PageIndex{4}\).44a), and an equilibrium between a monomer (Figure \(\PageIndex{4}\).44b) and dimer (Figure \(\PageIndex{4}\).44c) in the vapor phase.
| M | Structure |
|---|---|
| Be | Polymer (4-coordinate Be) |
| Mg | Cadmium chloride structure (6-coordinate Mg) |
| Ca | Deformed rutile structure (6-coordinate Ca) |
| Ca | Deformed rutile structure (6-coordinate Sr) |
| Ba | PbCl2 structure (9-coordinate Ba) or fluorite structure (8-coordinate Ba) |
Magnesium
The ionic radius for the +2 cation of magnesium is fairly small (0.65 Å). As a consequence the charge density (z/r) is high, which results in a high polarizing power of the Mg2+ ion. Thus, magnesium tends to form polar covalent bonds rather than ionic complexes. As with lithium there is a wide range of organometallic derivatives of magnesium, especially the Grignards (RMgX, where X = Cl, Br).
A further consequence of the covalent character of the bonding is that magnesium tends to form either 4-coordinate (tetrahedral) or 6-coordinate (octahedral) complexes with well-defined geometries.


