9.6: Discovery of Noble Gas Compounds
- Page ID
- 183351
In 1962 at the University of British Columbia, Neil Bartlett was working with the powerful oxidizer PtF6 and, because of an accidental leak in his vacuum line, noticed the compound’s reaction with O2 to generate a solid with formula "PtF6O2." The formula suggested Pt in the +10 oxidation state, which was clearly unreasonable because PtF6 was known to be a more powerful oxidizer than either molecular fluorine (F2) or molecular oxygen (O2). Bartlett noticed that the X-ray powder diffraction pattern of the compound was similar to that of Cs+AsF6-, a salt with the CsCl structure in which octahedral AsF6- ions occupy the chloride ion sites. This led Bartlett to propose a formulation of O2+PtF6- for his new compound.[6] Magnetic susceptibility data subsequently confirmed the presence of the paramagnetic O2+ cation, which (see Chapter 2) has a bond order of 2.5. This formulation implies that PtF6 was a strong enough oxidizing agent to oxidize molecular oxygen.
But just how strong an oxidizer is PtF6? Its electron affinity could be estimated by using a Born-Haber cycle, filling in the lattice energy of O2+PtF6- by means of Kapustinskii's formula:
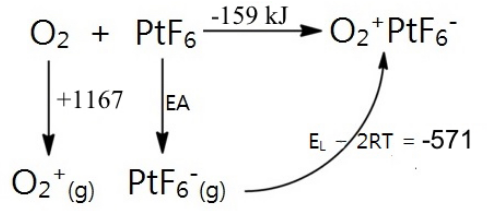
The electron affinity (EA) for PtF6 can be calculated as EA = -159 - 1167 + 571 = -751 kJ/mol. To put it in perspective, this is 417 kJ/mol more exothermic than the electron affinity of atomic fluorine (334 kJ). PtF6 was by far the strongest oxidizer that had ever been made!

Bartlett recognized that Xe has ionization energy of +1170 kJ, which is very close to the ionization energy of O2. Since Xe+ should be about the same size as O2+, the lattice energy should be about the same with Xe+ in the cation site of the O2+PtF6- structure. Since all of the other terms in the Born-Haber cycle for the reaction of Xe with PtF6 are the same, Bartlett concluded that Xe+PtF6-, like O2+PtF6-, should be a stable compound. He purchased a lecture bottle of xenon gas and reacted the two compounds, producing an orange solid.[7] While the product initially formed in the reaction may in fact be Xe+PtF6-, the Xe+ free radical is a powerful Lewis acid and reacts further with excess PtF6. The ultimate product of the reaction is formulated [XeF+][Pt2F11-], a salt which contains Xe in the +2 oxidation state and Pt in the +5 oxidation state. This was an important discovery because it shattered the dogmatic notion, which derived from the octet rule, that elements in group VIII could not form bonds with other elements. The name of this group was changed from the "inert gases" to the "noble gases." Subsequently, many compounds of Xe and a few of Kr and even Ar (which is much harder to oxidize) were synthesized and characterized.


