2.7: Periodic Trends in π Bonding
- Page ID
- 183302
As we noted in Section 2.3, pπ-bonding almost always involves a second-row element.
We encounter π-bonding from the sideways overlap of p-orbitals in the MO diagrams of second-row diatomics (B2…O2). It is important to remember that π-bonds are weaker than σ bonds made from the same AOs, and are especially weak if they involve elements beyond the second row.
Example:
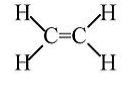 Ethylene: Stable molecule, doesn't polymerize without a catalyst.
Ethylene: Stable molecule, doesn't polymerize without a catalyst.
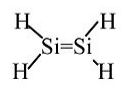 Silylene: Never isolated, spontaneously polymerizes. Calculations indicate 117 kJ/mol stability in the gas phase relative to singly-bonded (triplet) H2Si-SiH2.
Silylene: Never isolated, spontaneously polymerizes. Calculations indicate 117 kJ/mol stability in the gas phase relative to singly-bonded (triplet) H2Si-SiH2.
The large Ne core of Si atoms inhibits sideways overlap of 3p orbitals → weak π-bond.
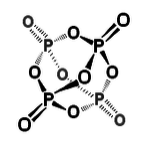
Other examples: P4 vs. N2
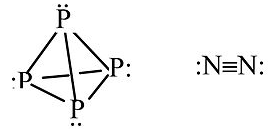
P cannot make π-bonds with itself, so it forms a tetrahedral molecule with substantial ring strain. This allotrope of P undergoes spontaneous combustion in air. Solid white phosphorus very slowly converts to red phosphorus, a more stable allotrope that contains sheets of pyramidal P atoms, each with bonds to three neighboring atoms and one lone pair.
|
White phosphorus (P4) is a soft, waxy solid that ignites spontaneously in air, burning with a bright flame and generating copious white P4O10 smoke. The sample shown here is photographed under water to prevent the oxidation reaction |
N can make π-bonds, so N2 has a very strong triple bond and is a relatively inert diatomic gas.
(CH3)2SiO vs. (CH3)2CO
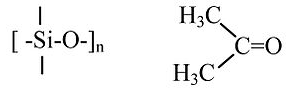
“RTV” silicone polymer (4 single bonds to Si) vs. acetone (C=O double bond). Silicones are soft, flexible polymers that can be heated to high temperatures (>300 °C) without decomposing. Acetone is a flammable molecular liquid that boils at 56 °C.
|
Silicone polymers (R2SiO)n are used in non-stick cookware like these muffin cups, in Silly Putty, soft robotics, and many other applications. |
Exceptions:
2nd row elements can form reasonably strong π-bonds with the smallest of the 3rd row elements, P, S, and Cl. Thus we find S=N bonds in sulfur-nitrogen compounds such as S2N2 and S3N3-, P=O bonds in phosphoric acid and P4O10 (shown above), and a delocalized π-molecular orbital in SO2 (as in ozone).




