7.E: Periodic Properties of the Elements (Exercises)
- Last updated
- Save as PDF
- Page ID
- 24787
These are homework exercises to accompany the Textmap created for "Chemistry: The Central Science" by Brown et al. Complementary General Chemistry question banks can be found for other Textmaps and can be accessed here. In addition to these publicly available questions, access to private problems bank for use in exams and homework is available to faculty only on an individual basis; please contact Delmar Larsen for an account with access permission.
7.1: Development of the Periodic Table
Conceptual Problems
- Johannes Dobereiner is credited with developing the concept of chemical triads. Which of the group 15 elements would you expect to compose a triad? Would you expect B, Al, and Ga to act as a triad? Justify your answers.
- Despite the fact that Dobereiner, Newlands, Meyer, and Mendeleev all contributed to the development of the modern periodic table, Mendeleev is credited with its origin. Why was Mendeleev’s periodic table accepted so rapidly?
- How did Moseley’s contribution to the development of the periodic table explain the location of the noble gases?
- The eka- naming scheme devised by Mendeleev was used to describe undiscovered elements.
- Use this naming method to predict the atomic number of eka-mercury, eka-astatine, eka-thallium, and eka-hafnium.
- Using the eka-prefix, identify the elements with these atomic numbers: 79, 40, 51, 117, and 121.
Numerical Problem
- Based on the data given, complete the table.
Species Molar Mass (g/mol) Density (g/cm3) Molar Volume (cm3/mol) A 40.078 25.85 B 39.09 0.856 C 32.065 16.35 D 1.823 16.98 E 26.98 9.992 F 22.98 0.968 Plot molar volume versus molar mass for these substances. According to Meyer, which would be considered metals and which would be considered nonmetals?
Numerical Answer
-
Species Molar Mass (g/mol) Density (g/cm3) Molar Volume (cm3/mol) A 40.078 1.550 25.85 B 39.09 0.856 45.67 C 32.065 1.961 16.35 D 30.95 1.823 16.98 E 26.98 2.700 9.992 F 22.98 0.968 23.7 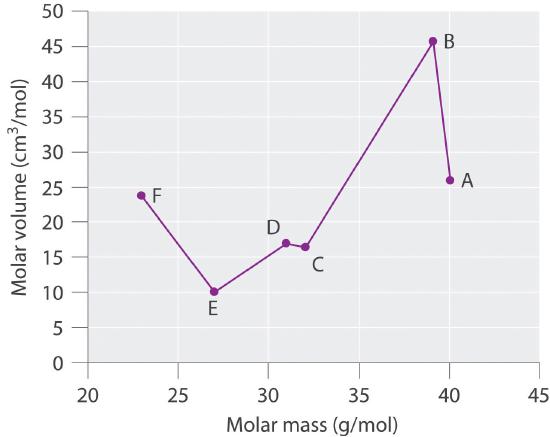
-
Meyer found that the alkali metals had the highest molar volumes, and that molar volumes decreased steadily with increasing atomic mass, then leveled off, and finally rose again. The elements located on the rising portion of a plot of molar volume versus molar mass were typically nonmetals. If we look at the plot of the data in the table, we can immediately identify those elements with the largest molar volumes (A, B, F) as metals located on the left side of the periodic table. The element with the smallest molar volume (E) is aluminum. The plot shows that the subsequent elements (C, D) have molar volumes that are larger than that of E, but smaller than those of A and B. Thus, C and D are most likely to be nonmetals (which is the case: C = sulfur, D = phosphorus).
7.2: Effective Nuclear Charge
Conceptual Problems
-
What happens to the energy of a given orbital as the nuclear charge Z of a species increases? In a multielectron atom and for a given nuclear charge, the Zeff experienced by an electron depends on its value of l. Why?
-
The electron density of a particular atom is divided into two general regions. Name these two regions and describe what each represents.
-
As the principal quantum number increases, the energy difference between successive energy levels decreases. Why? What would happen to the electron configurations of the transition metals if this decrease did not occur?
-
Describe the relationship between electron shielding and Zeff on the outermost electrons of an atom. Predict how chemical reactivity is affected by a decreased effective nuclear charge.
-
If a given atom or ion has a single electron in each of the following subshells, which electron is easier to remove?
- 2s, 3s
- 3p, 4d
- 2p, 1s
- 3d, 4s
7.3: Sizes of Atoms and Ions
Conceptual Problems
-
The electrons of the 1s shell have a stronger electrostatic attraction to the nucleus than electrons in the 2s shell. Give two reasons for this.
-
Predict whether Na or Cl has the more stable 1s2 shell and explain your rationale.
-
Arrange K, F, Ba, Pb, B, and I in order of decreasing atomic radius.
-
Arrange Ag, Pt, Mg, C, Cu, and Si in order of increasing atomic radius.
-
Using the periodic table, arrange Li, Ga, Ba, Cl, and Ni in order of increasing atomic radius.
-
Element M is a metal that forms compounds of the type MX2, MX3, and MX4, where X is a halogen. What is the expected trend in the ionic radius of M in these compounds? Arrange these compounds in order of decreasing ionic radius of M.
-
The atomic radii of Na and Cl are 190 and 79 pm, respectively, but the distance between sodium and chlorine in NaCl is 282 pm. Explain this discrepancy.
-
Are shielding effects on the atomic radius more pronounced across a row or down a group? Why?
-
What two factors influence the size of an ion relative to the size of its parent atom? Would you expect the ionic radius of S2− to be the same in both MgS and Na2S? Why or why not?
-
Arrange Br−, Al3+, Sr2+, F−, O2−, and I− in order of increasing ionic radius.
-
Arrange P3−, N3−, Cl−, In3+, and S2− in order of decreasing ionic radius.
-
How is an isoelectronic series different from a series of ions with the same charge? Do the cations in magnesium, strontium, and potassium sulfate form an isoelectronic series? Why or why not?
-
What isoelectronic series arises from fluorine, nitrogen, magnesium, and carbon? Arrange the ions in this series by
- increasing nuclear charge.
- increasing size.
-
What would be the charge and electron configuration of an ion formed from calcium that is isoelectronic with
- a chloride ion?
- Ar+?
Conceptual Answers
-
The 1s shell is closer to the nucleus and therefore experiences a greater electrostatic attraction. In addition, the electrons in the 2s subshell are shielded by the filled 1s2 shell, which further decreases the electrostatic attraction to the nucleus.
-
Ba > K > Pb > I > B > F
-
The sum of the calculated atomic radii of sodium and chlorine atoms is 253 pm. The sodium cation is significantly smaller than a neutral sodium atom (102 versus 154 pm), due to the loss of the single electron in the 3s orbital. Conversely, the chloride ion is much larger than a neutral chlorine atom (181 versus 99 pm), because the added electron results in greatly increased electron–electron repulsions within the filled n = 3 principal shell. Thus, transferring an electron from sodium to chlorine decreases the radius of sodium by about 50%, but causes the radius of chlorine to almost double. The net effect is that the distance between a sodium ion and a chloride ion in NaCl is greater than the sum of the atomic radii of the neutral atoms.
Numerical Problems
-
Plot the ionic charge versus ionic radius using the following data for Mo: Mo3+, 69 pm; Mo4+, 65 pm; and Mo5+, 61 pm. Then use this plot to predict the ionic radius of Mo6+. Is the observed trend consistent with the general trends discussed in the chapter? Why or why not?
-
Internuclear distances for selected ionic compounds are given in the following table.
-
If the ionic radius of Li+ is 76 pm, what is the ionic radius of each of the anions?
LiF LiCl LiBr LiI Distance (pm) 209 257 272 296
-
-
What is the ionic radius of Na+?
NaF NaCl NaBr NaI Distance (pm) 235 282 298 322 -
Arrange the gaseous species Mg2+, P3−, Br−, S2−, F−, and N3− in order of increasing radius and justify your decisions.
7.4: Ionization Energy
Conceptual Problems
-
Identify each statement as either true or false and explain your reasoning.
- Ionization energies increase with atomic radius.
- Ionization energies decrease down a group.
- Ionization energies increase with an increase in the magnitude of the electron affinity.
- Ionization energies decrease diagonally across the periodic table from He to Cs.
- Ionization energies depend on electron configuration.
- Ionization energies decrease across a row.
-
Based on electronic configurations, explain why the first ionization energies of the group 16 elements are lower than those of the group 15 elements, which is contrary to the general trend.
-
The first through third ionization energies do not vary greatly across the lanthanides. Why? How does the effective nuclear charge experienced by the ns electron change when going from left to right (with increasing atomic number) in this series?
-
Most of the first row transition metals can form at least two stable cations, for example iron(II) and iron(III). In contrast, scandium and zinc each form only a single cation, the Sc3+ and Zn2+ ions, respectively. Use the electron configuration of these elements to provide an explanation.
-
Of the elements Nd, Al, and Ar, which will readily form(s) +3 ions? Why?
-
Orbital energies can reverse when an element is ionized. Of the ions B3+, Ga3+, Pr3+, Cr3+, and As3+, in which would you expect this reversal to occur? Explain your reasoning.
-
The periodic trends in electron affinities are not as regular as periodic trends in ionization energies, even though the processes are essentially the converse of one another. Why are there so many more exceptions to the trends in electron affinities compared to ionization energies?
-
Elements lying on a lower right to upper left diagonal line cannot be arranged in order of increasing electronegativity according to where they occur in the periodic table. Why?
-
Why do ionic compounds form, if energy is required to form gaseous cations?
-
Why is Pauling’s definition of electronegativity considered to be somewhat limited?
-
Based on their positions in the periodic table, arrange Sb, O, P, Mo, K, and H in order of increasing electronegativity.
-
Based on their positions in the periodic table, arrange V, F, B, In, Na, and S in order of decreasing electronegativity.
Conceptual Answers
5. Both Al and Nd will form a cation with a +3 charge. Aluminum is in Group 13, and loss of all three valence electrons will produce the Al3+ ion with a noble gas configuration. Neodymium is a lanthanide, and all of the lanthanides tend to form +3 ions because the ionization potentials do not vary greatly across the row, and a +3 charge can be achieved with many oxidants.
11. K < Mo ≈ Sb < P ≈ H < O
Numerical Problems
-
The following table gives values of the first and third ionization energies for selected elements:
Number of Electrons Element I1 (E → E+ + e−, kJ/mol) Element I3 (E2+ → E3+ + e−, kJ/mol) 11 Na 495.9 Al 2744.8 12 Mg 737.8 Si 3231.6 13 Al 577.6 P 2914.1 14 Si 786.6 S 3357 15 P 1011.9 Cl 3822 16 S 999.6 Ar 3931 17 Cl 1251.2 K 4419.6 18 Ar 1520.6 Ca 4912.4 Plot the ionization energies versus the number of electrons. Explain why the slopes of the I1 and I3 plots are different, even though the species in each row of the table have the same electron configurations.
-
Would you expect the third ionization energy of iron, corresponding to the removal of an electron from a gaseous Fe2+ ion, to be larger or smaller than the fourth ionization energy, corresponding to the removal of an electron from a gaseous Fe3+ ion? Why? How would these ionization energies compare to the first ionization energy of Ca?
-
Which would you expect to have the highest first ionization energy: Mg, Al, or Si? Which would you expect to have the highest third ionization energy. Why?
-
Use the values of the first ionization energies given in Figure 7.11 to construct plots of first ionization energy versus atomic number for (a) boron through oxygen in the second period; and (b) oxygen through tellurium in group 16. Which plot shows more variation? Explain the reason for the variation in first ionization energies for this group of elements.
-
Arrange Ga, In, and Zn in order of increasing first ionization energies. Would the order be the same for second and third ionization energies? Explain your reasoning.
-
Arrange each set of elements in order of increasing magnitude of electron affinity.
- Pb, Bi, and Te
- Na, K, and Rb
- P, C, and Ge
-
Arrange each set of elements in order of decreasing magnitude of electron affinity.
- As, Bi, and N
- O, F, and Ar
- Cs, Ba, and Rb
-
Of the species F, O−, Al3+, and Li+, which has the highest electron affinity? Explain your reasoning.
-
Of the species O−, N2−, Hg2+, and H+, which has the highest electron affinity? Which has the lowest electron affinity? Justify your answers.
-
The Mulliken electronegativity of element A is 542 kJ/mol. If the electron affinity of A is −72 kJ/mol, what is the first ionization energy of element A? Use the data in the following table as a guideline to decide if A is a metal, a nonmetal, or a semimetal. If 1 g of A contains 4.85 × 1021 molecules, what is the identity of element A?
Na Al Si S Cl EA (kJ/mol) −59.6 −41.8 −134.1 −200.4 −348.6 I (kJ/mol) 495.8 577.5 786.5 999.6 1251.2 -
Based on their valence electron configurations, classify the following elements as either electrical insulators, electrical conductors, or substances with intermediate conductivity: S, Ba, Fe, Al, Te, Be, O, C, P, Sc, W, Na, B, and Rb.
-
Using the data in Problem 10, what conclusions can you draw with regard to the relationship between electronegativity and electrical properties? Estimate the approximate electronegativity of a pure element that is very dense, lustrous, and malleable.
-
Of the elements Al, Mg, O2, Ti, I2, and H2, which, if any, would you expect to be a good reductant? Explain your reasoning.
-
Of the elements Zn, B, Li, Se, Co, and Br2, which if any, would you expect to be a good oxidant? Explain your reasoning.
-
Determine whether each species is a good oxidant, a good reductant, or neither.
- Ba
- Mo
- Al
- Ni
- O2
- Xe
-
Determine whether each species is a good oxidant, a good reductant, or neither.
- Ir
- Cs
- Be
- B
- N
- Po
- Ne
-
Of the species I2, O−, Zn, Sn2+, and K+, choose which you would expect to be a good oxidant. Then justify your answer.
- Based on the valence electron configuration of the noble gases, would you expect them to have positive or negative electron affinities? What does this imply about their most likely oxidation states? their reactivity?
Numerical Answers
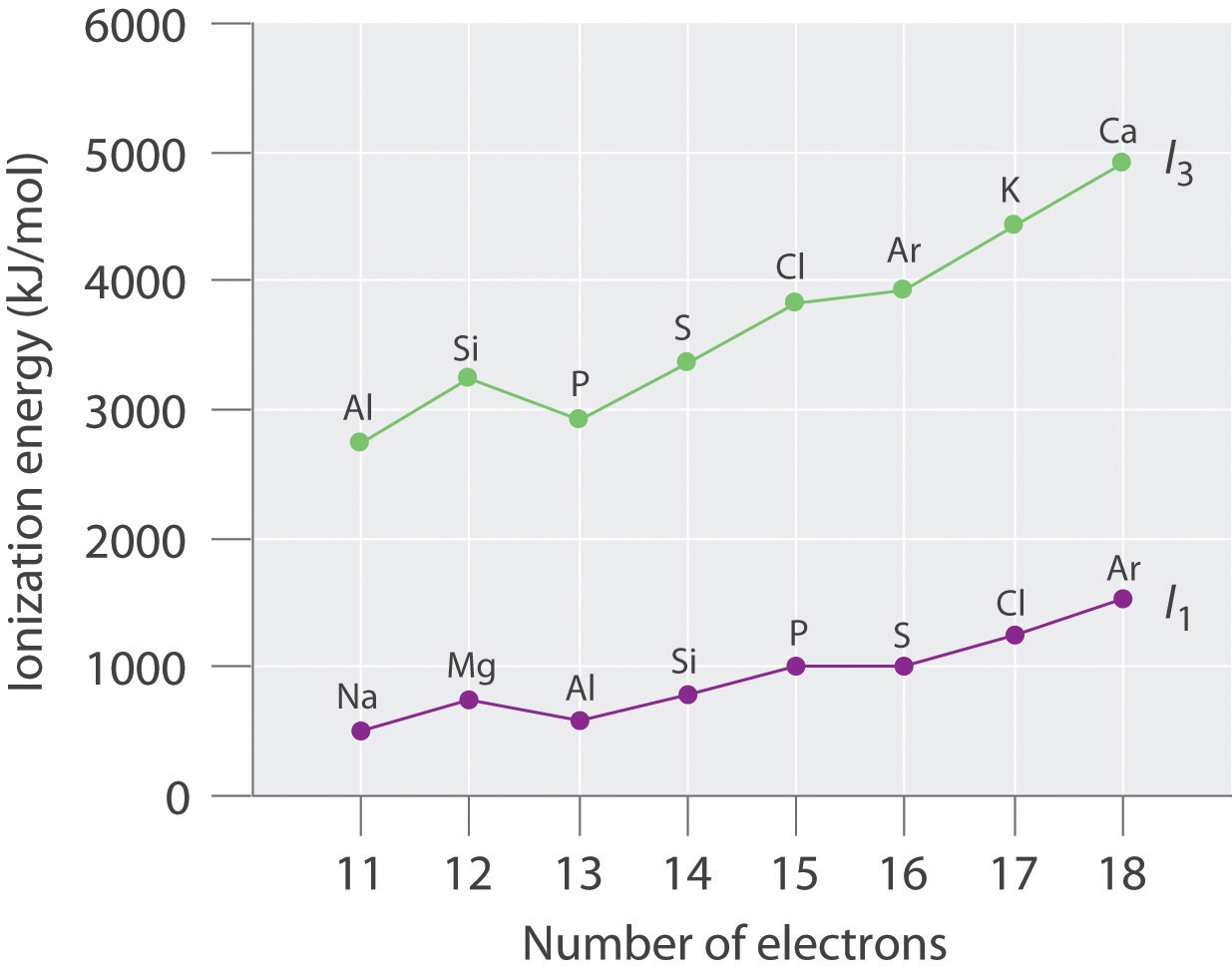
The general features of both plots are roughly the same, with a small peak at 12 electrons and an essentially level region from 15–16 electrons. The slope of the I3 plot is about twice as large as the slope of the I1 plot, however, because the I3 values correspond to removing an electron from an ion with a +2 charge rather than a neutral atom. The greater charge increases the effect of the steady rise in effective nuclear charge across the row.
-
-
Electron configurations: Mg, 1s22s22p63s2; Al, 1s22s22p63s23p1; Si, 1s22s22p63s23p2; First ionization energies increase across the row due to a steady increase in effective nuclear charge; thus, Si has the highest first ionization energy. The third ionization energy corresponds to removal of a 3s electron for Al and Si, but for Mg it involves removing a 2p electron from a filled inner shell; consequently, the third ionization energy of Mg is the highest.
-
- Bi > As > N
- F > O >> Ar
- Rb > Cs > Ba
- Hg2+ > H+ > O− > N2−; Hg2+ has the highest positive charge plus a relatively low energy vacant set of orbitals (the 6p subshell) to accommodate an added electron, giving it the greatest electron affinity; N2− has a greater negative charge than O−, so electron–electron repulsions will cause its electron affinity to be even lower (more negative) than that of O−.
-
-
insulators: S, O, C (diamond), P; conductors: Ba, Fe, Al, C (graphite), Be, Sc, W, Na, Rb; Te and B are semimetals and semiconductors.
-
-
Mg, Al, Ti, and H2
-
-
- reductant
- neither
- reductant
- reductant
- oxidant
- neither
-
-
I2 is the best oxidant, with a moderately strong tendency to accept an electron to form the I− ion, with a closed shell electron configuration. O− would probably also be an oxidant, with a tendency to add an electron to form salts containing the oxide ion, O2−. Zn and Sn2+ are all reductants, while K+ has no tendency to act as an oxidant or a reductant.

