7.6: Metals, Nonmetals, and Metalloids
- Page ID
- 21742
- To understand the basic properties separating Metals from Nonmetals and Metalloids
An element is the simplest form of matter that cannot be split into simpler substances or built from simpler substances by any ordinary chemical or physical method. There are 118 elements known to us, out of which 92 are naturally occurring, while the rest have been prepared artificially. Elements are further classified into metals, non-metals, and metalloids based on their properties, which are correlated with their placement in the periodic table.
| Metallic Elements | Nonmetallic elements |
|---|---|
| Distinguishing luster (shine) | Non-lustrous, various colors |
| Malleable and ductile (flexible) as solids | Brittle, hard or soft |
| Conduct heat and electricity | Poor conductors |
| Metallic oxides are basic, ionic | Nonmetallic oxides are acidic, covalent |
| Form cations in aqueous solution | Form anions, oxyanions in aqueous solution |
Metals
With the exception of hydrogen, all elements that form positive ions by losing electrons during chemical reactions are called metals. Thus metals are electropositive elements with relatively low ionization energies. They are characterized by bright luster, hardness, ability to resonate sound and are excellent conductors of heat and electricity. Metals are solids under normal conditions except for Mercury.
Physical Properties of Metals
Metals are lustrous, malleable, ductile, good conductors of heat and electricity. Other properties include:
- State: Metals are solids at room temperature with the exception of mercury, which is liquid at room temperature (Gallium is liquid on hot days).
- Luster: Metals have the quality of reflecting light from their surface and can be polished e.g., gold, silver and copper.
- Malleability: Metals have the ability to withstand hammering and can be made into thin sheets known as foils. For example, a sugar cube sized chunk of gold can be pounded into a thin sheet that will cover a football field.
- Ductility: Metals can be drawn into wires. For example, 100 g of silver can be drawn into a thin wire about 200 meters long.
- Hardness: All metals are hard except sodium and potassium, which are soft and can be cut with a knife.
- Valency: Metals typically have 1 to 3 electrons in the outermost shell of their atoms.
- Conduction: Metals are good conductors because they have free electrons. Silver and copper are the two best conductors of heat and electricity. Lead is the poorest conductor of heat. Bismuth, mercury and iron are also poor conductors
- Density: Metals have high density and are very heavy. Iridium and osmium have the highest densities whereas lithium has the lowest density.
- Melting and Boiling Points: Metals have high melting and boiling points. Tungsten has the highest melting and boiling points whereas mercury has the lowest. Sodium and potassium also have low melting points.
Chemical Properties of Metals
Metals are electropositive elements that generally form basic or amphoteric oxides with oxygen. Other chemical properties include:
- Electropositive Character: Metals tend to have low ionization energies, and typically lose electrons (i.e. are oxidized) when they undergo chemical reactions They normally do not accept electrons. For example:
- Alkali metals are always 1+ (lose the electron in s subshell)
- Alkaline earth metals are always 2+ (lose both electrons in s subshell)
- Transition metal ions do not follow an obvious pattern, 2+ is common (lose both electrons in s subshell), and 1+ and 3+ are also observed
\[\ce{Na^0 \rightarrow Na^+ + e^{-}} \label{1.1} \]
\[\ce{Mg^0 \rightarrow Mg^{2+} + 2e^{-}} \label{1.2} \]
\[\ce{Al^0 \rightarrow Al^{3+} + 3e^{-}} \label{1.3} \]
Compounds of metals with non-metals tend to be ionic in nature. Most metal oxides are basic oxides and dissolve in water to form metal hydroxides:
\[\ce{Na2O(s) + H2O(l) \rightarrow 2NaOH(aq)}\label{1.4} \]
\[\ce{CaO(s) + H2O(l) \rightarrow Ca(OH)2(aq)} \label{1.5} \]
Metal oxides exhibit their basic chemical nature by reacting with acids to form metal salts and water:
\[\ce{MgO(s) + HCl(aq) \rightarrow MgCl2(aq) + H2O(l)} \label{1.6} \]
\[\ce{NiO(s) + H2SO4(aq) \rightarrow NiSO4(aq) + H2O(l)} \label{1.7} \]
What is the chemical formula for aluminum oxide?
Solution
Al has a 3+ charge, the oxide ion is \(O^{2-}\), thus \(Al_2O_3\).
Would you expect it to be solid, liquid or gas at room temperature?
Solutions
Oxides of metals are characteristically solid at room temperature
Write the balanced chemical equation for the reaction of aluminum oxide with nitric acid:
Solution
Metal oxide + acid -> salt + water
\[\ce{Al2O3(s) + 6HNO3(aq) \rightarrow 2Al(NO3)3(aq) + 3H2O(l)} \nonumber \]
Nonmetals
Elements that tend to gain electrons to form anions during chemical reactions are called non-metals. These are electronegative elements with high ionization energies. They are non-lustrous, brittle and poor conductors of heat and electricity (except graphite). Non-metals can be gases, liquids or solids.
Physical Properties of Nonmetals
- Physical State: Most of the non-metals exist in two of the three states of matter at room temperature: gases (oxygen) and solids (carbon). Only bromine exists as a liquid at room temperature.
- Non-Malleable and Ductile: Non-metals are very brittle, and cannot be rolled into wires or pounded into sheets.
- Conduction: They are poor conductors of heat and electricity.
- Luster: These have no metallic luster and do not reflect light.
- Melting and Boiling Points: The melting points of non-metals are generally lower than metals, but are highly variable.
- Seven non-metals exist under standard conditions as diatomic molecules: \(\ce{H2(g)}\), \(\ce{N2(g)}\), \(\ce{O2(g)}\), \(\ce{F2(g)}\), \(\ce{Cl2(g)}\), \(\ce{Br2(l)}\), \(\ce{I2(s)}\).
Chemical Properties of Nonmetals
Non-metals have a tendency to gain or share electrons with other atoms. They are electronegative in character. Nonmetals, when reacting with metals, tend to gain electrons (typically attaining noble gas electron configuration) and become anions:
\[\ce{3Br2(l) + 2Al(s) \rightarrow 2AlBr3(s)} \nonumber \]
Compounds composed entirely of nonmetals are covalent substances. They generally form acidic or neutral oxides with oxygen that that dissolve in water to form acids:
\[\ce{CO2(g) + H2O(l)} \rightarrow \underset{\text{carbonic acid}}{\ce{H2CO3(aq)}} \nonumber \]
As you may know, carbonated water is slightly acidic (carbonic acid).
Nonmetal oxides can combine with bases to form salts.
\[\ce{CO2(g) + 2NaOH(aq) \rightarrow Na2CO3(aq) + H2O(l)} \nonumber \]
Metalloids
Metalloids have properties intermediate between the metals and nonmetals. Metalloids are useful in the semiconductor industry. Metalloids are all solid at room temperature. They can form alloys with other metals. Some metalloids, such as silicon and germanium, can act as electrical conductors under the right conditions, thus they are called semiconductors. Silicon for example appears lustrous, but is not malleable nor ductile (it is brittle - a characteristic of some nonmetals). It is a much poorer conductor of heat and electricity than the metals. The physical properties of metalloids tend to be metallic, but their chemical properties tend to be non-metallic. The oxidation number of an element in this group can range from +5 to -2, depending on the group in which it is located.
| Metals | Non-metals | Metalloids |
|---|---|---|
| Gold | Oxygen | Silicon |
| Silver | Carbon | Boron |
| Copper | Hydrogen | Arsenic |
| Iron | Nitrogen | Antimony |
| Mercury | Sulfur | Germanium |
| Zinc | Phosphorus |
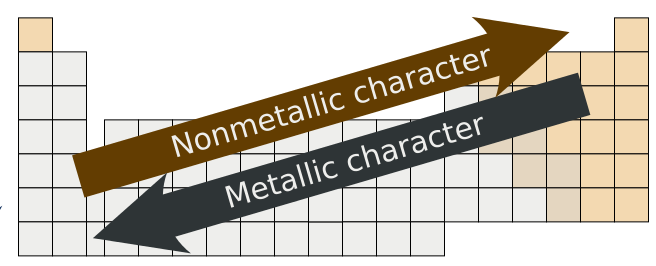
Trends in Metallic and Nonmetallic Character
Metallic character is strongest for the elements in the leftmost part of the periodic table, and tends to decrease as we move to the right in any period (nonmetallic character increases with increasing electronegativity and ionization energy values). Within any group of elements (columns), the metallic character increases from top to bottom (the electronegativity and ionization energy values generally decrease as we move down a group). This general trend is not necessarily observed with the transition metals.
Contributors and Attributions
Binod Shrestha (University of Lorraine)

