2.E: Atoms, Molecules, and Ions (Exercises)
- Page ID
- 43465
These are homework exercises to accompany the Textmap created for "Chemistry: The Central Science" by Brown et al. Complementary General Chemistry question banks can be found for other Textmaps and can be accessed here. In addition to these publicly available questions, access to private problems bank for use in exams and homework is available to faculty only on an individual basis; please contact Delmar Larsen for an account with access permission.
2.1: The Atomic Theory of Matter
Conceptual Problems
-
Which of the following elements exist as diatomic molecules?
- helium
- hydrogen
- iodine
- gold
-
Which of the following elements exist as diatomic molecules?
- chlorine
- potassium
- silver
- oxygen
-
Why is it proper to represent the elemental form of helium as He but improper to represent the elemental form of hydrogen as H?
-
Why is it proper to represent the elemental form of chlorine as Cl2 but improper to represent the elemental form of calcium as Ca2?
Conceptual Solutions
- Hydrogen and Iodine.
- Chlorine and Oxygen.
- Hydrogen exists as a diatomic molecule in its elemental form; helium does not exist as a diatomic molecule.
- Chlorine exists naturally in its diatomic form, while Calcium does not.
Exercises
- What is the modern atomic theory?
- What are atoms?
Conceptual Answers
- The modern atomic theory states that all matter is composed of atoms, which are unique between elements. They cannot be created or destroyed, but can combine with one another in whole number ratios to form compounds.
- Atoms are the smallest parts of an element that maintain the identity of that element.
Numerical Problems
- (Basic concept check) When 32.0 grams (g) of methane are burned in 128.0 g of oxygen, 88.0 g of carbon dioxide and 72.0 g of water are produced. Which law is this an example of? (a) Law of definite proportions (b) Law of conservation of mass or (c) Law of multiple proportions.
- (Law of Conservation of Mass) 8.00 grams (g) of methane are burned in 32.00 g of oxygen. The reaction produces 22.00 g of carbon dioxide and an unmeasured mass of water. What mass of water is produced?
- (Law of Definite Proportions) Two experiments using sodium and chlorine are performed. In the first experiment, 4.36 grams (g) sodium are reacted with 32.24 g of chlorine, using up all the sodium. 11.08 g of sodium chloride was produced in the first experiment. In the second experiment, 4.20 g of chlorine reacted with 20.00 g of sodium, using up all the chlorine. 6.92 g of sodium chloride was produced in the second experiment. Show that these results are consistent with the law of constant composition.
- (Law of Conservation of Mass): 36.0 grams (g) of wood are burned in oxygen. The products of this reaction weigh 74.4 g. (a) What mass of oxygen is needed in this reaction? (b) What mass of oxygen is needed to burn 8.00 lb of wood? 1 lb = 453.59237 g.
- (Law of Definite Proportions): A sample of methane contains only carbon and hydrogen, with 3.00 grams (g) of carbon for every 1.00 g of hydrogen. How much hydrogen should be present in a different, 50.0 g same of methane?
Numerical Solutions
- The answer is (b) Law of conservation of mass. The number of grams of reactants (32.0 g of methane and 128.0 g of oxygen = 160.0 g total) is equal to the number of grams of product (88.0 g of carbon dioxide and 72.0 g of water = 160.0 g total).
- The answer is 18.00 g of water. Because the only products are water and carbon dioxide, their total mass must equal the total mass of the reactants, methane and oxygen. 8.00 g of methane + 32.00 g of oxygen = 40.00 total g of reactants. Because the total mass of the reactants equals the total mass of the products, the total mass of the products is also 40.00 g. Thus, 40.00 total g of products = 22.00 g carbon dioxide + unknown mass water. 40.00 total g of products - 22.00 g carbon dioxide = 18.00 g water.
-
To solve, determine the percent of sodium in each sample of sodium chloride. There is 4.36 g sodium for every 11.08 g of sodium chloride in the first experiment. The amount of sodium in the sodium chloride for the second experiment must be found. This is found by subtracted the known amount of reacted chlorine (4.20 g) from the amount of sodium chloride (6.92 g). 6.92 g sodium chloride - 4.20 g chlorine = 2.72 g sodium.
Thus, the percent of sodium in each sample is represented below:
% Na = (4.36 g Na)/(11.08 g NaCl) x 100% = 39.4% Na % Na = (2.72 g Na)/(6.92 g NaCl) x 100% = 39.3%
The slight difference in compositions is due to significant figures: each percent has an uncertainty of .01% in either direction. The two samples of sodium chloride have the same composition. -
- The answer is 38.4 g of oxygen. The total mass of the products is 74.4 g. Thus, the total mass of the reactants must equal 74.4 g as well. Thus, 74.4 g products - 36.0 g wood reactant = 38.4 g oxygen reactant.
- The answer is 8.53 lb of oxygen. From, (a) that it takes 38.4 g of oxygen to burn 18.0 g of wood. First, convert both of these values to pounds (alternatively, the 8.00 lb can be converted to grams).
36.0 g wood x (1lb)/(453.59237 g) = 0.0793664144 lb wood
38.4 g oxygen x (1 lb)/(453.59237 g) = .0846575087 lb oxygen
Now two ratios equal to each other can be set up to determine the unknown mass of oxygen.
(0.0793664144 lb wood)/(.0846575087 lb oxygen) = (8.00 lb wood)/(unknown mass oxygen)
Solving reveals that it requires 8.53 lb of oxygen to burn 8.00 lb of wood.
- The answer is 12.5 g of hydrogen. If there are 3.00 g of carbon present for every 1.00 g of hydrogen, we can assume the smallest whole number combination of these elements in that ratio to be 4.00 g of methane:
50.0 g methane x (1.00 g hydrogen)/(4.00 g methane) = 12.5 g of hydrogen.
2.3: The Modern View of Atomic Structure
Conceptual Problems
- Describe the experiment that provided evidence that the proton is positively charged.
- What observation led Rutherford to propose the existence of the neutron?
- What is the difference between Rutherford’s model of the atom and the model chemists use today?
- If cathode rays are not deflected when they pass through a region of space, what does this imply about the presence or absence of a magnetic field perpendicular to the path of the rays in that region?
- Describe the outcome that would be expected from Rutherford’s experiment if the charge on α particles had remained the same but the nucleus were negatively charged. If the nucleus were neutral, what would have been the outcome?
- Describe the differences between an α particle, a β particle, and a γ ray. Which has the greatest ability to penetrate matter?
Conceptual Solutions
- Ernest Rutherford's 1911 Alpha-ray scattering experiment proved that the nucleus of an atom is positively charged. In the experiment, he fired high-energy streams of α particles at a thin gold sheet. Many passed through unimpeded, but some were deflected slightly and few were reflected back, proving that the positive charge of an atom is contained in the nucleus.
- Rutherford proposed the existence of the neutron when discovering that electrons and protons alone did not fully account for the mass of the atom, leading him to work with James Chadwick to prove the existence of a neutral particle.
- While Rutherford's model established the positively charged nucleus surrounded by negatively charged electrons, it stated that the electrons were evenly distributed throughout, rather than in "specific energy levels" of electrons, which are a significant part of the modern model.
- In the absence of any other fields affecting the cathode ray, a lack of deflection implies an absence of a magnetic field in a direction that would affect their path.
- If the nucleus were negatively charged, one could expect α particles to be deflected towards the nucleus rather than away. If it were neutral, it would have no effect on the particles.
- α particles are helium nuclei (effectively He ions lacking electrons), β particle are fast-moving electrons, and γ rays are very short electromagnetic rays. Out of the three, γ rays have the greatest penetrative ability.
Numerical Problems
Please be sure you are familiar with the topics discussed in Section 1.6 before proceeding to the Numerical Problems.
- Using the data in Table 1.3 and the periodic table, calculate the percentage of the mass of a silicon atom that is due to
- electrons.
- protons.
- Using the data in Table 1.3 and the periodic table, calculate the percentage of the mass of a helium atom that is due to
- electrons.
- protons.
- The radius of an atom is approximately 104 times larger than the radius of its nucleus. If the radius of the nucleus were 1.0 cm, what would be the radius of the atom in centimeters? in miles?
- The total charge on an oil drop was found to be 3.84 × 10−18 coulombs. What is the total number of electrons contained in the drop?
Numerical Solutions
- The total atomic mass of a silicon atom is 28.085amu. Using the data from the table (as well as silicon's atomic number of 14), we can calculate that the mass of electrons in the atom is 0.0076804amu, while the mass of protons is 14.101864amu. From this we can calculate that electrons take up 0.02735% of the mass of the atom, while protons take up 50.2114% of the atom's mass.
- The total atomic mass of a helium atom is 4.0026amu. Using the data from the table we see that the mass of electrons in the atom is 0.0010972amu, while the mass of protons is 2.015amu. Therefore, electrons take up 0.0274% of the mass of the atom, while protons take up 50.342%.
- If the nucleus were to have a radius of 1.0cm, the atom would have a radius of 10000cm, or 0.06 miles.
- The electrical charge of a single electron is equal to -1.602 × 10-19. Thus, we can divide the total charge of the oil drop by this number to see that there are 24 electrons contained within it.
2.4: Atomic Mass
Conceptual Problems
1. Complete the following table for the missing elements, symbols, and numbers of electrons.
| Element | Symbol | Number of Electrons |
| molybdenum | ||
| 19 | ||
| titanium | ||
| B | ||
| 53 | ||
| Sm | ||
| helium | ||
| 14 |
2. Complete the following table for the missing elements, symbols, and numbers of electrons.
| Element | Symbol | Number of Electrons |
| lanthanum | ||
| Ir | ||
| aluminum | ||
| 80 | ||
| sodium | ||
| Si | ||
| 9 | ||
| Be |
3. Is the mass of an ion the same as the mass of its parent atom? Explain your answer.
4. What isotopic standard is used for determining the mass of an atom?
5. Give the symbol \(^A_Z X\) for these elements, all of which exist as a single isotope.
- beryllium
- ruthenium
- phosphorus
- aluminum
- cesium
- praseodymium
- cobalt
- yttrium
- arsenic
6. Give the symbol \(_Z^AX\) for these elements, all of which exist as a single isotope.
- fluorine
- helium
- terbium
- iodine
- gold
- scandium
- sodium
- niobium
- manganese
7. Identify each element, represented by X, that has the given symbols.
- \(_{26}^{55}X\)
- \(_{33}^{74}X\)
- \(_{12}^{24}X\)
- \(_{53}^{127}X\)
- \(_{18}^{40}X\)
- \(_{63}^{152}X\)
Conceptual Solutions
-
Element Symbol Number of Electrons molybdenum Mo 42 potassium K 19 titanium Ti 22 boron B 5 iodine I 53 samarium Sm 62 helium He 2 silicon Si 14 -
Element Symbol Number of Electrons lanthanum La 57 iridium Ir 77 aluminum Al 13 mercury Hg 80 sodium Na 11 silicon Si 14 flourine F 9 beryllium Be 4 - Although the mass of an ion is not exactly the same as its parent atom, electrons weigh so little that removing them will not change the mass in any significant way. Thus, we can say that the mass of an ion is the same.
- The reference isotope which is used for determining the mass of all other atoms is the carbon-12 nuclide, which is set to weigh exactly 12 amu. Therefore other elements are massed in relation to this value.
- Give the symbol \(^A_Z X\) for these elements, all of which exist as a single isotope.
- beryllium
- \(^9_4 Be\)
- ruthenium
- \(^{102}_{44} Ru\)
- phosphorus
- \(^{31}_{15} P\)
- aluminum
- \(^{27}_{13} Al\)
- cesium
- \(^{133}_{55} Cs\)
- praseodymium
- \(^{141}_{59} Pr\)
- cobalt
- \(^{59}_{27} Co\)
- yttrium
- \(^{89}_{39} Y\)
- arsenic
- \(^{75}_{33} As\)
- beryllium
-
Give the symbol \(_Z^AX\) for these elements.
-
fluorine
-
\(^{19}_{9} F\)
-
-
helium
-
\(^{4}_{2} He\)
-
- terbium
- \(^{159}_{65} Tb\)
- iodine
- \(^{127}_{53} I\)
- gold
- \(^{197}_{79} Au\)
- scandium
- \(^{45}_{21} Sc\)
- sodium
- \(^{23}_{11} Na\)
- niobium
- \(^{93}_{41} Nb\)
- manganese
- \(^{55}_{25} Mn\)
-
-
Identify each element, represented by X, that has the given symbols.
- \(_{26}^{55}X\)
- Iron (Fe)
- \(_{33}^{74}X\)
- Arsenic (As)
- \(_{12}^{24}X\)
- Magnesium (Mg)
- \(_{53}^{127}X\)
- Iodine (I)
- \(_{18}^{40}X\)
- Argon (Ar)
- \(_{63}^{152}X\)
- Europium (Eu)
- \(_{26}^{55}X\)
Numerical Problems
Please be sure you are familiar with the topics discussed in Section 1.6 before proceeding to the Numerical Problems.
1. The isotopes 131I and 60Co are commonly used in medicine. Determine the number of neutrons, protons, and electrons in a neutral atom of each.
2. Determine the number of protons, neutrons, and electrons in a neutral atom of each isotope:
- \(^{97}Tc\)
- \(^{113}In\)
- \(^{63}Ni\)
- \(^{55}Fe\)
3. Both technetium-97 and americium-240 are produced in nuclear reactors. Determine the number of protons, neutrons, and electrons in the neutral atoms of each.
4. The following isotopes are important in archeological research. How many protons, neutrons, and electrons does a neutral atom of each contain?
- \(^{207}Pb\)
- \(^{16}O\)
- \(^{40}K\)
- \(^{137}Cs\)
- \(^{40}Ar\)
5. Copper, an excellent conductor of heat, has two isotopes: 63Cu and 65Cu. Use the following information to calculate the average atomic mass of copper:
| Isotope | Percent Abundance (%) | Atomic Mass (amu) |
| 63Cu | 69.09 | 62.9298 |
| 65Cu | 30.92 | 64.9278 |
6. Silicon consists of three isotopes with the following percent abundance:
| Isotope | Percent Abundance (%) | Atomic Mass (amu) |
| 28Si | 92.18 | 27.976926 |
| 29Si | 4.71 | 28.976495 |
| 30Si | 3.12 | 29.973770 |
Calculate the average atomic mass of silicon.
7. Complete the following table for neon. The average atomic mass of neon is 20.1797 amu.
| Isotope | Percent Abundance (%) | Atomic Mass (amu) |
| 20Ne | 90.92 | 19.99244 |
| 21Ne | 0.257 | 20.99395 |
| 22Ne |
8. Are \(_{28}^{63} X \) and \( _{29}^{62} X \) isotopes of the same element? Explain your answer.
9.Complete the following table:
| Isotope | Number of Protons | Number of Neutrons | Number of Electrons |
| 238X | 95 | ||
| 238U | |||
| 75 | 112 |
10.Complete the following table:
| Isotope | Number of Protons | Number of Neutrons | Number of Electrons |
| 57Fe | |||
| 40X | 20 | ||
| 36S |
11. Using a mass spectrometer, a scientist determined the percent abundances of the isotopes of sulfur to be 95.27% for 32S, 0.51% for 33S, and 4.22% for 34S. Use the atomic mass of sulfur from the periodic table (see Chapter 32 "Appendix H: Periodic Table of Elements") and the following atomic masses to determine whether these data are accurate, assuming that these are the only isotopes of sulfur: 31.972071 amu for 32S, 32.971459 amu for 33S, and 33.967867 amu for 34S.
12. The percent abundances of two of the three isotopes of oxygen are 99.76% for 16O, and 0.204% for 18O. Use the atomic mass of oxygen given in the periodic table and the following data to determine the mass of 17O: 15.994915 amu for 16O and 17.999160 amu for 18O.
13. Which element has the higher proportion by mass in NaI?
14. Which element has the higher proportion by mass in KBr?
Numerical Solutions
- Determine the number of neutrons, protons, and electrons in a neutral atom of:
- 131I
- 53 Protons, 53 Electrons, 78 Neutrons. In a neutral atom, the amount of protons is equal to the amount of electrons. Additionally, the amount of neutrons can be found by taking [Mass Number] - [Amount of Protons].
- 60Co
- 27 Protons, 27 Electrons, 33 Neutrons.
- 131I
-
Determine the number of protons, neutrons, and electrons in a neutral atom of each isotope:
- \(^{97}Tc\)
- 43 Protons, 43 Electrons, 54 Neutrons
- \(^{113}In\)
- 49 Protons, 49 Electrons, 64 Neutrons
- \(^{63}Ni\)
- 28 Protons, 28 Electrons, 35 Neutrons
- \(^{55}Fe\)
- 26 Protons, 26 Electrons, 29 Neutrons
- \(^{97}Tc\)
- Determine the number of protons, neutrons, and electrons in the neutral atoms of each.
- Technetium-97
- 43 Protons, 43 Electrons, 54 Neutrons
- Americium-240
- 95 Protons, 95 Electrons, 145 Neutrons
- Technetium-97
-
How many protons, neutrons, and electrons does a neutral atom of each contain?
- \(^{207}Pb\)
- 82 Protons, 82 Electrons, 125 Neutrons
- \(^{16}O\)
- 8 Protons, 8 Electrons, 8 Neutrons
- \(^{40}K\)
- 19 Protons, 19 Electrons, 21 Neutrons
- \(^{137}Cs\)
- 55 Protons, 55 Electrons, 82 Neutrons
- \(^{40}Ar\)
- 18 Protons, 18 Electrons, 22 Neutrons
- \(^{207}Pb\)
- The average atomic mass of an element from its isotopes can be found via the equation: Average A. Mass = f1M1 + f2M2 +… + fnMn, where f is the abundance of an isotope and M is that isotope's mass. For example, for the given table of copper, we can plug in (0.6909 × 62.9298) + (0.3092 × 64.9278) = 63.5539amu.
- Average Atomic Mass of Silicon: (0.9218 × 27.796926) + (0.0471 × 28.976495) + (0.0312 × 29.97377) = 27.9232amu.
-
Isotope Percent Abundance (%) Atomic Mass (amu) 20Ne 90.92 19.99244 21Ne 0.257 20.99395 22Ne 8.823 22.08568 - They are not isotopes of the same element, because the number of protons (and therefore the atomic number) are different. For two isotopes to be of the same element, they must share an atomic number.
-
Isotope Number of Protons Number of Neutrons Number of Electrons 238X 95 143 95 238U 92 146 92 187Re 75 112 75 -
Isotope Number of Protons Number of Neutrons Number of Electrons 57Fe 26 31 26 40X 20 20 20 36S 16 20 16 - To solve this problem, we will use the same method we did for problem 6. With the values given we can plug them into the formula to obtain (0.9572 × 31.9720) + (0.0051 × 32.9715) + (0.0422 × 33.9679) = 32.2052amu. The periodic table states that the mass of sulfur is 32.06amu, meaning that the measurements are not entirely accurate.
- From the given percent abundances, we can calculate that the abundance of 17O is 0.036%. Plugging this into the formula we can solve for the mass of 17O, which is 15.985amu. Obviously, it does not make much sense for an isotope of oxygen containing more neutrons to be lighter, meaning the data is not entirely correct.
- Iodine has a higher proportion by mass in NaI. For every 22.99amu of Na, there are 126.9amu of I.
- Bromine has a higher proportion by mass in KBr. For every 39.098amu of K, there are 79.904amu of Br.
2.5: The Periodic Table
Conceptual Problems
1. Classify each element in Conceptual Problem 1 of section 2.4 as a metal, a nonmetal, or a semimetal. If a metal, state whether it is an alkali metal, an alkaline earth metal, or a transition metal.
2. Classify each element in Conceptual Problem 2 of section 2.4 as a metal, a nonmetal, or a semimetal. If a metal, state whether it is an alkali metal, an alkaline earth metal, or a transition metal.
3. Classify each element as a metal, a semimetal, or a nonmetal. If a metal, state whether it is an alkali metal, an alkaline earth metal, or a transition metal.
- iron
- tantalum
- sulfur
- silicon
- chlorine
- nickel
- potassium
- radon
- zirconium
4. Which of these sets of elements are all in the same period?
- potassium, vanadium, and ruthenium
- lithium, carbon, and chlorine
- sodium, magnesium, and sulfur
- chromium, nickel, and krypton
5. Which of these sets of elements are all in the same period?
- barium, tungsten, and argon
- yttrium, zirconium, and selenium
- potassium, calcium, and zinc
- scandium, bromine, and manganese
6. Which of these sets of elements are all in the same group?
- sodium, rubidium, and barium
- nitrogen, phosphorus, and bismuth
- copper, silver, and gold
- magnesium, strontium, and samarium
7. Which of these sets of elements are all in the same group?
- iron, ruthenium, and osmium
- nickel, palladium, and lead
- iodine, fluorine, and oxygen
- boron, aluminum, and gallium
8. Indicate whether each element is a transition metal, a halogen, or a noble gas.
- manganese
- iridium
- fluorine
- xenon
- lithium
- carbon
- zinc
- sodium
- tantalum
- hafnium
- antimony
- cadmium
9. Which of the elements indicated in color in the periodic table shown below is most likely to exist as a monoatomic gas? As a diatomic gas? Which is most likely to be a semimetal? A reactive metal?
10. Based on their locations in the periodic table, would you expect these elements to be malleable? Why or why not?
- phosphorus
- chromium
- rubidium
- copper
- aluminum
- bismuth
- neodymium
11. Based on their locations in the periodic table, would you expect these elements to be lustrous? Why or why not?
- sulfur
- vanadium
- nickel
- arsenic
- strontium
- cerium
- sodium
Conceptual Solution
-
Element Symbol Type molybdenum Mo metal; transition metal potassium K metal; alkali metal titanium Ti metal; transition metal boron B semimetal iodine I nonmetal samarium Sm metal; lanthanide helium He nonmetal silicon Si semimetal -
Element Symbol Type lanthanum La metal; lanthanide iridium Ir metal; transition metal aluminum Al metal; post-transition metal mercury Hg metal; transition metal sodium Na metal; alkali metal silicon Si semimetal flourine F nonmetal beryllium Be metal; alkaline earth metal -
Symbol Type Fe metal: transition metal Ta metal: transition metal S nonmetal Si semimetal Cl nonmetal (halogen) Ni metal: transition metal K metal: alkali metal Rn nonmetal (noble gas) Zr metal: transition metal - C and D. All elements in set C are in the 3rd period, and all elements in set D are in the 4th period.
- C and D. All elements in both of these sets are in the 4th period.
- B and C. All elements in set B are in group 15, while all elements in set C are in group 11.
- A and D. All elements in set A are in group 8, and all elements in set D are in group 13.
-
Element Symbol Type manganese Mn transition metal iridium Ir transition metal flourine F halogen xenon Xe noble gas lithium Li alkali metal carbon C nonmetal zinc Zn transition metal sodium Na alkali metal tantalum Ta transition metal hafnium Hf transition metal antimony Sb semimetal cadmium Cd transition metal - The red element is most likely to exist as a monoatomic gas, as it is a noble gas meaning that all electrons are paired, and it has no need to bond with itself. The yellow element is most likely to exist as a diatomic gas, as it is in the halogen group meaning it has one unpaired electron. The orange element is most likely to be a semimetal, being within both the group and period ranges that include semimetals. The green element is most likely to be a reactive metal, as it is in the first group making it an alkali metal.
- Note that though some elements are expected to be malleable based on their position, that is not always the case. For example, Bismuth would be expected to be malleable due to its status as a post-transition metal, but it is in fact brittle.
-
Element Expected to be Malleable? Why? phosphorus No Reactive Nonmetal chromium Yes Transition Metal rubidium Yes Alkali Metal copper Yes Transition Metal aluminium Yes Post-Transition Metal bismuth Yes Post-Transition Metal neodymium Yes Lanthanoid (Metal)
-
-
Element Lustrous? Why? sulfur No Reactive Nonmetal vanadium Yes Transition Metal nickel Yes Transition Metal arsenic Yes Metalloid strontium Yes Alkaline Earth Metal cerium Yes Lanthanoid (Metal) sodium Yes Alkali Metal
2.6: Molecules and Molecular Compounds
Conceptual Problems
- Ionic and covalent compounds are held together by electrostatic attractions between oppositely charged particles. Describe the differences in the nature of the attractions in ionic and covalent compounds. Which class of compounds contains pairs of electrons shared between bonded atoms?
- Which contains fewer electrons than the neutral atom—the corresponding cation or the anion?
- What is the difference between an organic compound and an inorganic compound?
- What is the advantage of writing a structural formula as a condensed formula?
- The majority of elements that exist as diatomic molecules are found in one group of the periodic table. Identify the group.
- Discuss the differences between covalent and ionic compounds with regard to
- a. the forces that hold the atoms together.
- b. melting points.
- c. physical states at room temperature and pressure.
- Why do covalent compounds generally tend to have lower melting points than ionic compounds?
Conceptual Answer
- Ionic compounds are held together by powerful electromagnetic forces, usually caused by a more electronegative atom "taking" an electron from another to become an ion. Covalent compounds, however, share electrons between them in a steady balance between attractive and repulsive charges.
- Cations, being positively charged, possess fewer electrons than their neutral atoms.
- Organic compounds involve Carbon and Hydrogen in nearly all cases, while inorganic compounds consist of the other elements.
- Condensed formulas can be written out more quickly and easily, and are helpful when showing that multiple atoms are connected to a single one in a compound.
- Group 17, Halogens
-
Covalent Ionic Forces Electrostatic Attraction between Nuclei and Electrons Electrostatic Attraction between Cations and Anions Melting Points Very Low Very High Physical States at STP Liquid/Gas Solid
7. Covalent compounds generally melt at lower temperatures than ionic compounds because the intermolecular interactions that hold the molecules together in a molecular solid are weaker than the electrostatic attractions that hold oppositely charged ions together in an ionic solid.
Numerical Problems
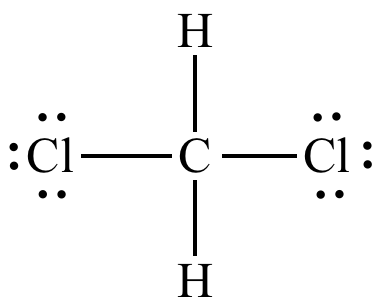
1. The structural formula for chloroform (CHCl3) was shown in Example 2.6.2. Based on this information, draw the structural formula of dichloromethane (CH2Cl2).
2. What is the total number of electrons present in each ion?
- F−
- Rb+
- Ce3+
- Zr4+
- Zn2+
- Kr2+
- B3+
3. What is the total number of electrons present in each ion?
- Ca2+
- Se2−
- In3+
- Sr2+
- As3+
- N3−
- Tl+
4. Predict how many electrons are in each ion.
- an oxygen ion with a −2 charge
- a beryllium ion with a +2 charge
- a silver ion with a +1 charge
- a selenium ion with a +4 charge
- an iron ion with a +2 charge
- a chlorine ion with a −1 charge
5. Predict how many electrons are in each ion.
- copper ion with a +2 charge
- a molybdenum ion with a +4 charge
- an iodine ion with a −1 charge
- a gallium ion with a +3 charge
- an ytterbium ion with a +3 charge
- a scandium ion with a +3 charge
6. Predict the charge on the most common monatomic ion formed by each element.
- chlorine
- phosphorus
- scandium
- magnesium
- arsenic
- oxygen
7. Predict the charge on the most common monatomic ion formed by each element.
- sodium
- selenium
- barium
- rubidium
- nitrogen
- aluminum
8. For each representation of a monatomic ion, identify the parent atom, write the formula of the ion using an appropriate superscript, and indicate the period and group of the periodic table in which the element is found.
- \(_4^9X^{2+} \)
- \(_1^1X^-\)
- \(_8^{16}X^{2-} \)
9. For each representation of a monatomic ion, identify the parent atom, write the formula of the ion using an appropriate superscript, and indicate the period and group of the periodic table in which the element is found.
- \(_3^7X^+ \)
- \(_9^{19}X^-\)
- \(_{13}^{27}X^{3+}\)
Numerical Answers
- Total number of electrons present in each ion:
- F−
- 10
- Rb+
- 36
- Ce3+
- 55
- Zr4+
- 36
- Zn2+
- 28
- Kr2+
- 34
- B3+
- 2
- F−
- Total number of electrons present in each ion:
- Ca2+
- 18
- Se2−
- 36
- In3+
- 46
- Sr2+
- 36
- As3+
- 30
- N3−
- 10
- Tl+
- 80
- Ca2+
- How many electrons are in each ion:
- an oxygen ion with a −2 charge
- 10
- a beryllium ion with a +2 charge
- 2
- a silver ion with a +1 charge
- 46
- a selenium ion with a +4 charge
- 30
- an iron ion with a +2 charge
- 24
- a chlorine ion with a −1 charge
- 18
- an oxygen ion with a −2 charge
-
How many electrons are in each ion:
- copper ion with a +2 charge
- 27
- a molybdenum ion with a +4 charge
- 38
- an iodine ion with a −1 charge
- 54
- a gallium ion with a +3 charge
- 28
- an ytterbium ion with a +3 charge
- 67
- a scandium ion with a +3 charge
- 18
- copper ion with a +2 charge
-
The charge on the most common monatomic ion formed by each element:
- chlorine
- -1
- phosphorus
- -3
- scandium
- +3
- magnesium
- +2
- arsenic
- -3
- oxygen
- -2
- chlorine
-
The charge on the most common monatomic ion formed by each element:
- sodium
- +1
- selenium
- -2
- barium
- +2
- rubidium
- +1
- nitrogen
- +3
- aluminum
- +3
- sodium
- Identify the parent atom, write the formula of the ion using an appropriate superscript, and indicate the period and group of the periodic table in which the element is found.
- \(_4^9X^{2+} \)
- Parent Atom
- Be
- Formula of Ion
- Be2+
- Period and Group
- Period 2, Group 2
- Parent Atom
- \(_1^1X^-\)
- Parent Atom
- H
- Formula of Ion
- H-
- Period and Group
- Period 1, Group 1
- Parent Atom
- \(_8^{16}X^{2-} \)
- Parent Atom
- O
- Formula of Ion
- O2-
- Period and Group
- Period 2, Group 16
- Parent Atom
- \(_4^9X^{2+} \)
- Identify the parent atom, write the formula of the ion using an appropriate superscript, and indicate the period and group of the periodic table in which the element is found.
- \(_3^7X^+ \)
- Parent Atom
- Li
- Formula of Ion
- Li+
- Period and Group
- Period 2, Group 1
- Parent Atom
- \(_9^{19}X^-\)
- Parent Atom
- F
- Formula of Ion
- F-
- Period and Group
- Period 2, Group 17
- Parent Atom
- \(_{13}^{27}X^{3+}\)
- Parent Atom
- Al
- Formula of Ion
- Al3+
- Period and Group
- Period 3, Group 13
- Parent Atom
- \(_3^7X^+ \)
2.8: Naming Inorganic Compounds
Conceptual Problems
1. What are the differences and similarities between a polyatomic ion and a molecule?
2. Classify each compound as ionic or covalent.
- Zn3(PO4)2
- C6H5CO2H
- K2Cr2O7
- CH3CH2SH
- NH4Br
- CCl2F2
3. Classify each compound as ionic or covalent. Which are organic compounds and which are inorganic compounds?
- CH3CH2CO2H
- CaCl2
- Y(NO3)3
- H2S
- NaC2H3O2
4. Generally, one cannot determine the molecular formula directly from an empirical formula. What other information is needed?
5. Give two pieces of information that we obtain from a structural formula that we cannot obtain from an empirical formula.
6. The formulas of alcohols are often written as ROH rather than as empirical formulas. For example, methanol is generally written as CH3OH rather than CH4O. Explain why the ROH notation is preferred.
7. The compound dimethyl sulfide has the empirical formula C2H6S and the structural formula CH3SCH3. What information do we obtain from the structural formula that we do not get from the empirical formula? Write the condensed structural formula for the compound.
8. What is the correct formula for magnesium hydroxide—MgOH2 or Mg(OH)2? Why?
9. Magnesium cyanide is written as Mg(CN)2, not MgCN2. Why?
10. Does a given hydrate always contain the same number of waters of hydration?
Conceptual Solutions
- Polyatomic ions and molecules are similar in that both are groups of multiple atoms held together by covalent bonds. However, polyatomic ions have net electrical charges, either positive or negative, while molecules do not.
-
Classify each compound as ionic or covalent.
- Zn3(PO4)2
- Ionic
- C6H5CO2H
- Covalent
- K2Cr2O7
- Ionic
- CH3CH2SH
- Covalent
- NH4Br
- Covalent
- CCl2F2
- Covalent
- Zn3(PO4)2
- Classify each compound as ionic or covalent, organic or inorganic.
- CH3CH2CO2H
- Covalent, organic
- CaCl2
- Ionic, inorganic
- Y(NO3)3
- Ionic, inorganic
- H2S
- Covalent, inorganic
- NaC2H3O2
- Ionic, organic
- CH3CH2CO2H
- In order to find the molecular formula from the empirical formula, the molar mass of the compound is also needed.
- Structural Formulas allow us to see the types of bonds connecting atoms within a molecule or ion (single, double, or triple bonds), as well as a general shape in which the atoms are arranged.
- OH is the functional group of the alcohol family, which confers upon it its characteristic properties. By using the ROH notation, one can tell with a single glance that a compound is an alcohol.
- The structural formula gives us the connectivity of the atoms in the molecule or ion, as well as a schematic representation of their arrangement in space. Empirical formulas tell us only the ratios of the atoms present. The condensed structural formula of dimethylsulfide is (CH3)2S.
- The correct formula is Mg(OH)2. Being a neutral molecule, the charges on the components must even out to 0 — Mg has a charge of +2, O has a charge of -2, while H has a charge of +1. MgOH2 would result in a polyatomic ion with charge +2, since there would only be one O. Additionally, Hydroxide refers specifically to the diatomic anion OH-, so Magnesium Hydroxide would need to be composed of OH- due to the name.
- The functional group present in cyanides is the cyano group, which is CN. The cyano group has a charge of -1. In order for the molecule to be a Cyanide, it must contain this functional group, and since this is a neutral molecule the charges must add up to 0. Given that Mg has a charge of +2, there must be two CN groups in order to even out the charges.
- Yes. Each specific hydrate has a certain ratio of waters of hydration bound to it, differing between compounds (but not within the same hydrate).
Numerical Problems
1. Write the formula for each compound.
- magnesium sulfate, which has 1 magnesium atom, 4 oxygen atoms, and 1 sulfur atom
- ethylene glycol (antifreeze), which has 6 hydrogen atoms, 2 carbon atoms, and 2 oxygen atoms
- acetic acid, which has 2 oxygen atoms, 2 carbon atoms, and 4 hydrogen atoms
- potassium chlorate, which has 1 chlorine atom, 1 potassium atom, and 3 oxygen atoms
- sodium hypochlorite pentahydrate, which has 1 chlorine atom, 1 sodium atom, 6 oxygen atoms, and 10 hydrogen atoms
2. Write the formula for each compound.
- cadmium acetate, which has 1 cadmium atom, 4 oxygen atoms, 4 carbon atoms, and 6 hydrogen atoms
- barium cyanide, which has 1 barium atom, 2 carbon atoms, and 2 nitrogen atoms
- iron(III) phosphate dihydrate, which has 1 iron atom, 1 phosphorus atom, 6 oxygen atoms, and 4 hydrogen atoms
- manganese(II) nitrate hexahydrate, which has 1 manganese atom, 12 hydrogen atoms, 12 oxygen atoms, and 2 nitrogen atoms
- silver phosphate, which has 1 phosphorus atom, 3 silver atoms, and 4 oxygen atoms
3. Complete the following table by filling in the formula for the ionic compound formed by each cation-anion pair.
| Ion | K+ | Fe3+ | NH4+ | Ba2+ |
|---|---|---|---|---|
| Cl− | KCl | |||
| SO42− | ||||
| PO43− | ||||
| NO3− | ||||
| OH− |
4. Write the empirical formula for the binary compound formed by the most common monatomic ions formed by each pair of elements.
- zinc and sulfur
- barium and iodine
- magnesium and chlorine
- silicon and oxygen
- sodium and sulfur
5. Write the empirical formula for the binary compound formed by the most common monatomic ions formed by each pair of elements.
- lithium and nitrogen
- cesium and chlorine
- germanium and oxygen
- rubidium and sulfur
- arsenic and sodium
6. Write the empirical formula for each compound.
- Na2S2O4
- B2H6
- C6H12O6
- P4O10
- KMnO4
7. Write the empirical formula for each compound.
- Al2Cl6
- K2Cr2O7
- C2H4
- (NH2)2CNH
- CH3COOH
Numerical Answers
- Formula of each compound:
- magnesium sulfate
- MgSO4
- ethylene glycol (antifreeze)
- (CH2OH)2
- acetic acid
- CH₃COOH
- potassium chlorate
- KClO3
- sodium hypochlorite pentahydrate
- NaOCl·5H2O
- magnesium sulfate
- Formula of each compound:
- cadmium acetate, which has 1 cadmium atom, 4 oxygen atoms, 4 carbon atoms, and 6 hydrogen atoms
- Cd(O2CCH3)2
- barium cyanide, which has 1 barium atom, 2 carbon atoms, and 2 nitrogen atoms
- Ba(CN)2
- iron(III) phosphate dihydrate, which has 1 iron atom, 1 phosphorus atom, 6 oxygen atoms, and 4 hydrogen atoms
- FePO4 · 2H2O
- manganese(II) nitrate hexahydrate, which has 1 manganese atom, 12 hydrogen atoms, 12 oxygen atoms, and 2 nitrogen atoms
- Mn(NO3)2 · 6H2O
- silver phosphate, which has 1 phosphorus atom, 3 silver atoms, and 4 oxygen atoms
- Ag3PO4
- cadmium acetate, which has 1 cadmium atom, 4 oxygen atoms, 4 carbon atoms, and 6 hydrogen atoms
-
Ion K+ Fe3+ NH4+ Ba2+ Cl− KCl FeCl3 NH4Cl BaCl2 SO42− K2SO4 Fe2(SO4)3 (NH4)2SO4 BaSO4 PO4 3− K3PO4 FePO4 (NH4)3PO4 Ba3(PO4)2 NO3− KNO3 Fe(NO3)3 NH4NO3 Ba(NO3)2 OH− KOH Fe(OH)3 NH4OH Ba(OH)2 - The empirical formula for the binary compound:
- zinc and sulfur
- ZnS
- barium and iodine
- BaI2
- magnesium and chlorine
- MgCl2
- silicon and oxygen
- SiO2
- sodium and sulfur
- Na2S
- zinc and sulfur
- The empirical formula for the binary compound:
- lithium and nitrogen
- Li3N
- cesium and chlorine
- CsCl
- germanium and oxygen
- GeO2
- rubidium and sulfur
- Rb2S
- arsenic and sodium
- Na3As
- lithium and nitrogen
-
The empirical formula for each compound:
- Na2S2O4
- NaSO2
- B2H6
- BH3
- C6H12O6
- CH2O
- P4O10
- P2O5
- KMnO4
- KMnO4
- Na2S2O4
- The empirical formula for each compound:
- Al2Cl6
- AlCl3
- K2Cr2O7
- K2Cr2O7
- C2H4
- CH2
- (NH2)2CNH
- CH5N3
- CH3COOH
- CH2O
- Al2Cl6
2.9: Some Simple Organic Compounds
Conceptual Problems
1. Benzene (C6H6) is an organic compound, and KCl is an ionic compound. The sum of the masses of the atoms in each empirical formula is approximately the same. How would you expect the two to compare with regard to each of the following? What species are present in benzene vapor?
- melting point
- type of bonding
- rate of evaporation
- structure
2. Can an inorganic compound be classified as a hydrocarbon? Why or why not?
3. Is the compound NaHCO3 a hydrocarbon? Why or why not?
4. Name each compound.
- NiO
- TiO2
- N2O
- CS2
- SO3
- NF3
- SF6
5. Name each compound.
- HgCl2
- IF5
- N2O5
- Cl2O
- HgS
- PCl5
6. For each structural formula, write the condensed formula and the name of the compound.
a.
b.
c.
d.
e.
7. For each structural formula, write the condensed formula and the name of the compound.
a.
b.
c.
d.
8. Would you expect PCl3 to be an ionic compound or a covalent compound? Explain your reasoning.
9. What distinguishes an aromatic hydrocarbon from an aliphatic hydrocarbon?
10. The following general formulas represent specific classes of hydrocarbons. Refer to Table 2.7 "The First 10 Straight-Chain Alkanes" and Table 2.8 "Some Common Acids That Do Not Contain Oxygen" and Figure 2.16 and identify the classes.
- CnH2n + 2
- CnH2n
- CnH2n − 2
11. Using R to represent an alkyl or aryl group, show the general structure of an
- alcohol.
- phenol.
Conceptual Answer
-
Benzene KCl Melting Point Lower Higher Type of Bonding Covalent Ionic Rate of Evaporation Higher Lower Structure Planar Molecule Ionic Crystal - An inorganic compound cannot be classified as a hydrocarbon, as hydrocarbons are by definition organic.
- NaHCO3 is not a hydrocarbon. Hydrocarbons consist entirely of carbon and hydrogen atoms, while NaHCO3 also contains nitrogen and oxygen.
- Name each compound.
- NiO
- Nickel Oxide
- TiO2
- Titanium Dioxide
- N2O
- Nitrous Oxide
- CS2
- Carbon Disulfide
- SO3
- Sulfur Trioxide
- NF3
- Nitrogen Trifluoride
- SF6
- Sulfur Hexafluoride
- NiO
- Name each compound.
- HgCl2
- Mercury(II) Chloride
- IF5
- Iodine Pentafluoride
- N2O5
- Dinitrogen Pentoxide
- Cl2O
- Dichlorine Monoxide
- HgS
- Mercury(II) Sulfide
- PCl5
- Phosphorus Pentachloride
- HgCl2
- PCl3 would be expected to be a covalent compound. The electronegativity difference between P and Cl is not great enough to create an ionic bond.
- An aromatic hydrocarbon contains a conjugated ring system (in other words, a ring system composed of alternating single and double bonds). An aliphatic hydrocarbon, on the other hand, can be linear, or it can contain a non-conjugated ring system.
- Identify the classes.
- CnH2n + 2
- Alkane
- CnH2n
- Alkene
- CnH2n − 2
- Alkyne
- CnH2n + 2
-
- ROH, where R is an alkyl group, in other words an alkane where one hydrogen is removed. The OH is attached to a saturated carbon atom.
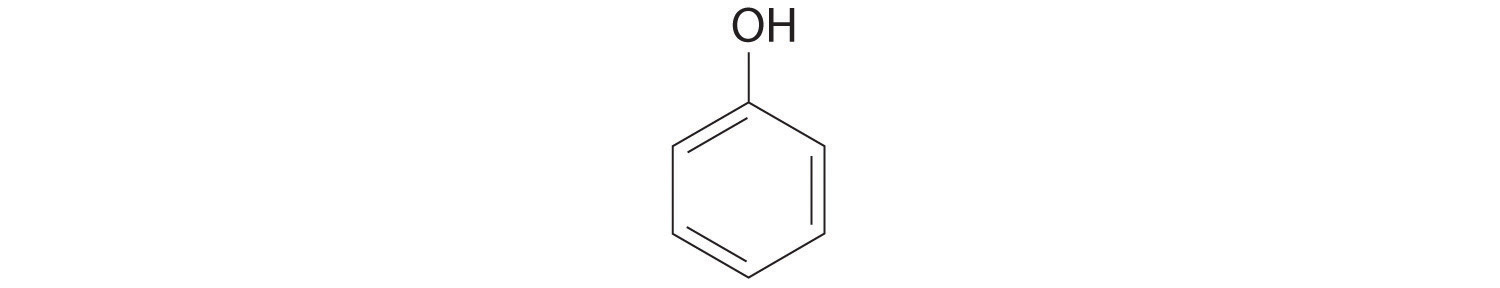
- ROH, where R is an aryl group, in other words an aromatic ring where one hydrogen is removed. The OH is attached directly to the ring.
Numerical Problems
1. Write the formula for each compound.
- dinitrogen monoxide
- silicon tetrafluoride
- boron trichloride
- nitrogen trifluoride
- phosphorus tribromide
2. Write the formula for each compound.
- dinitrogen trioxide
- iodine pentafluoride
- boron tribromide
- oxygen difluoride
- arsenic trichloride
3. Write the formula for each compound.
- thallium(I) selenide
- neptunium(IV) oxide
- iron(II) sulfide
- copper(I) cyanide
- nitrogen trichloride
4. Name each compound.
- RuO4
- PbO2
- MoF6
- Hg2(NO3)2·2H2O
- WCl4
5. Name each compound.
- NbO2
- MoS2
- P4S10
- Cu2O
- ReF5
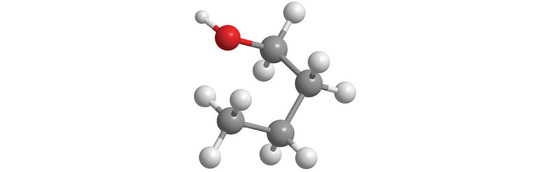
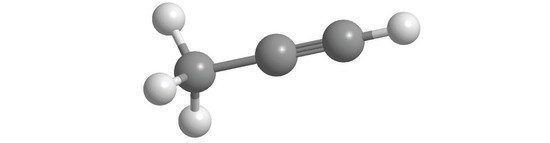
6. Draw the structure of each compound.
- propyne
- ethanol
- n-hexane
- cyclopropane
- benzene
7. Draw the structure of each compound.
- 1-butene
- 2-pentyne
- cycloheptane
- toluene
- phenol
Numerical Answers
-
- N2O
- SiF4
- BCl3
- NF3
- PBr3
-
- N2O3
- IF5
- BBr3
- OF2
- AsCl3
-
- TlSe
- NpO2
- FeS
- CuCN
- NCl3
-
- Ruthenium Tetroxide
- Lead Dioxide (or Lead(IV) Oxide)
- Molybdenum Hexafluoride (or Molybdenum(VI) Fluoride)
- Mercury(I) Nitrate Dihydrate
- Tungsten Tetrachloride (or Tungsten(IV) Chloride)
- niobium (IV) oxide
- molybdenum (IV) sulfide
- tetraphosphorus decasulfide
- copper(I) oxide
- rhenium(V) fluoride
6.
7.
a.
b.
c.
d.
e.