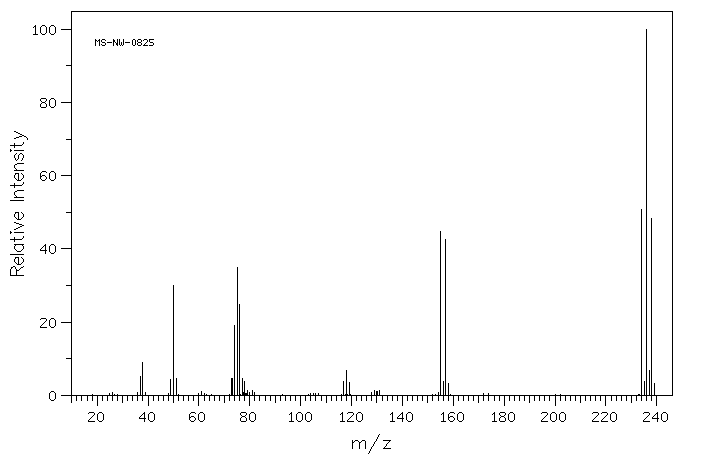
6.7: Other Important Isotopes- Br and Cl
- Page ID
- 195146
There are other cases in which knowledge of isotopes can be crucial. For example, bromoethane should display a peak for the molecular ion at m/z = 109. However, the farthest large peak to the right in its mass spectrum is at 110. There is another large peak at 108.

Source: SDBSWeb : http://riodb01.ibase.aist.go.jp/sdbs/ (National Institute of Advanced Industrial Science and Technology of Japan, 22 August 2008)
- In the periodic table, the atomic mass of bromine is listed as 80, but that is just an average.
- Bromine is really about 50% 79Br and 50% 81Br.
- As a result, two molecular ion peaks of equal intensity two units apart, M+ and M+2, are observed.
- This pattern of molecular ions is a good indication that there is a bromine present in the molecule.
Similarly, chlorobenzene should display M+ at m/z =112, provided you take into account that most chlorine atoms have an atomic weight of 35 amu. The periodic table lists an atomic weight for chlorine of 35.453 amu, though. That's because about 25% of chlorine atoms are actually 37Cl. The mass spectrum of chlorobenzene actually shows an additional molecular ion at 114 amu.
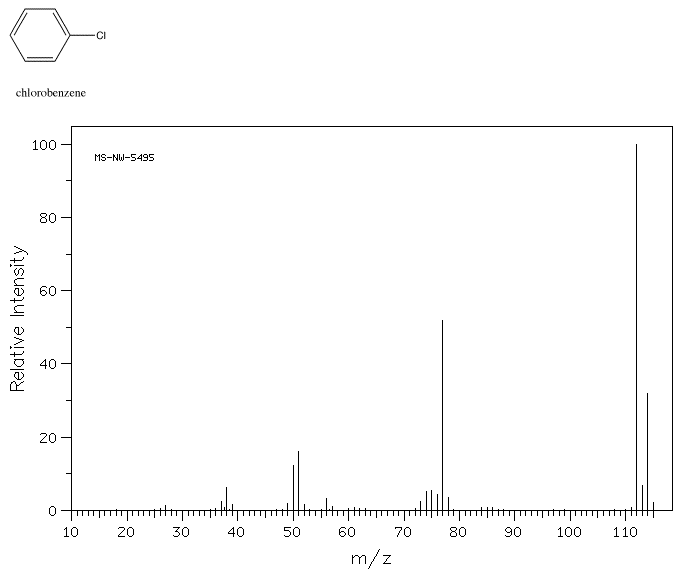
Source: SDBSWeb : http://riodb01.ibase.aist.go.jp/sdbs/ (National Institute of Advanced Industrial Science and Technology of Japan, 22 August 2008)
- Chlorinated compounds show an M+2 peak that is 1/3 as large as the M+ peak.
Note also that halogens are easily lost during mass spectrometry. If you subtract the mass of the halogen from the molecular ion mass, you will often find a peak that corresponds to the remainder of the structure.
Draw one possible structure for the compound in each of the following mass spectra.
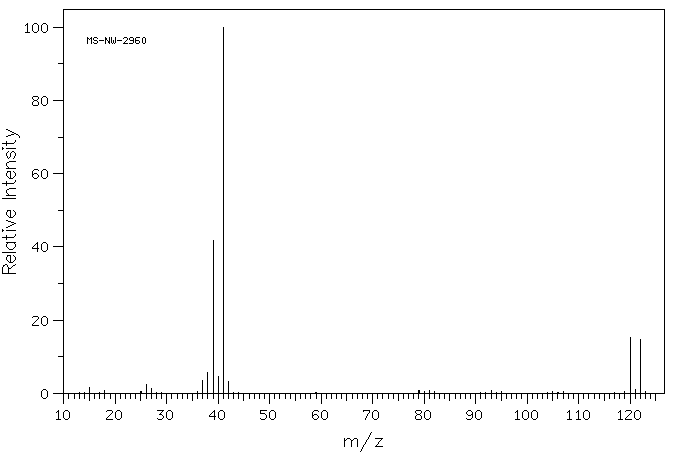
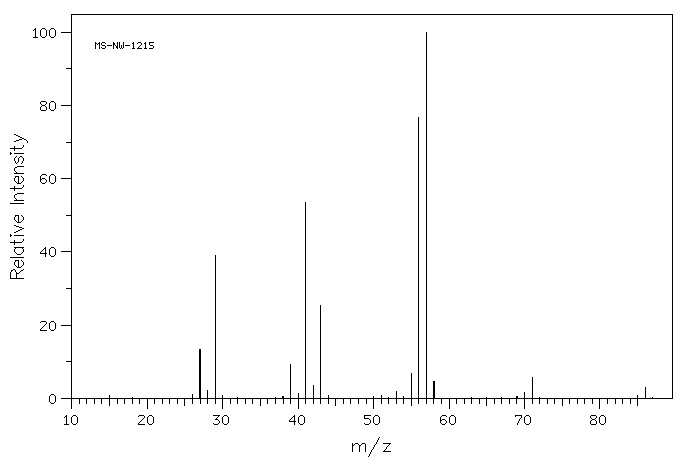
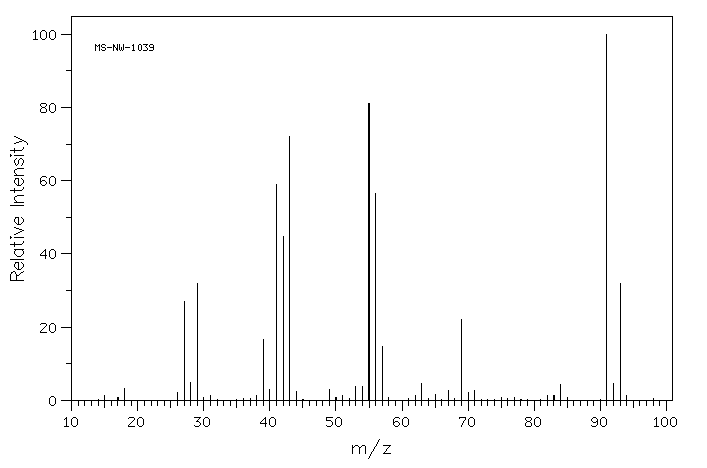
In the following mass spectrum, more than one halogen atom is present.
- What is a possible structure of the compound?
- Show why this pattern of molecular ions is observed.