3.10: Additional Problems
- Page ID
- 194823
For each of the following hand-drawn cartoons of IR spectra, identify the bond(s) that correspond to the indicated peaks.
i.
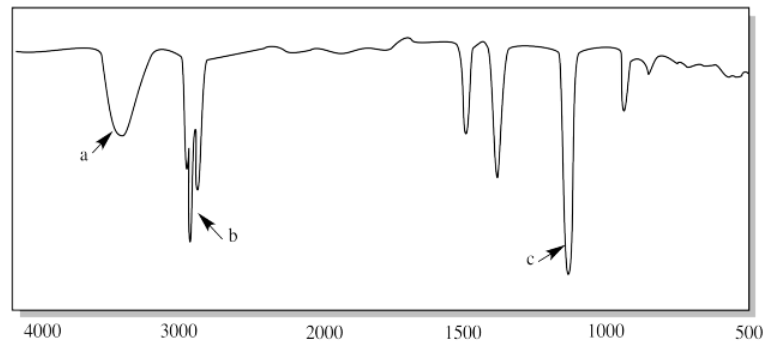
ii.
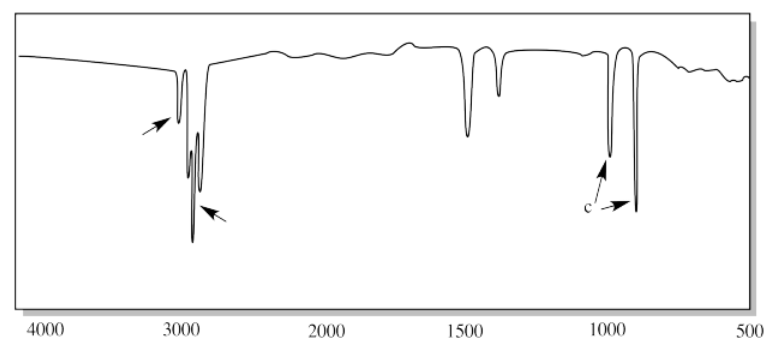
iii.
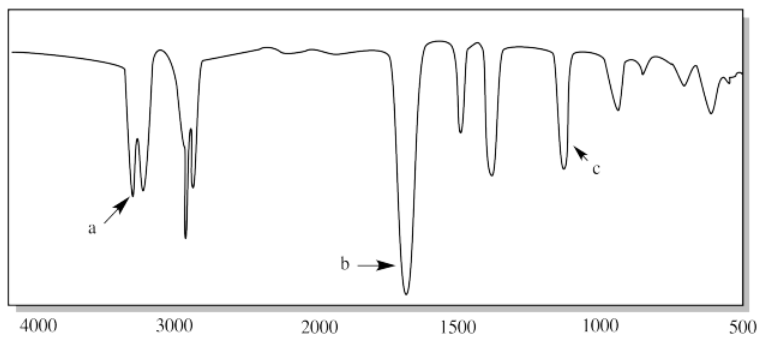
iv.
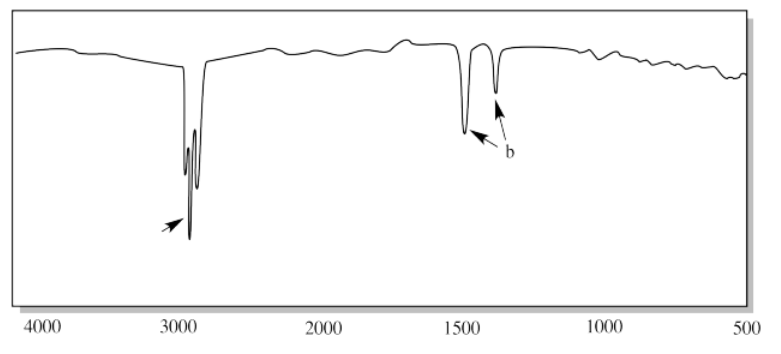
v.
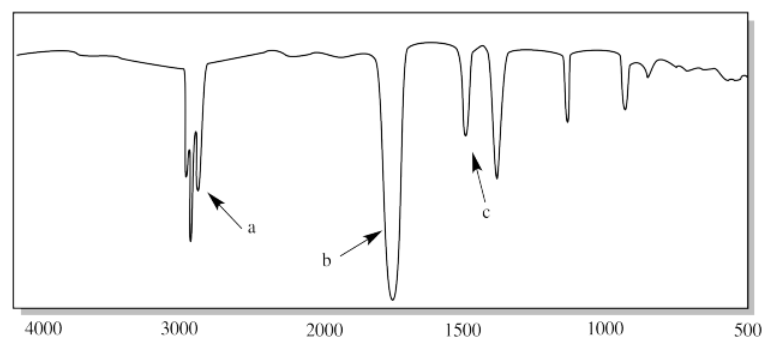
vi.
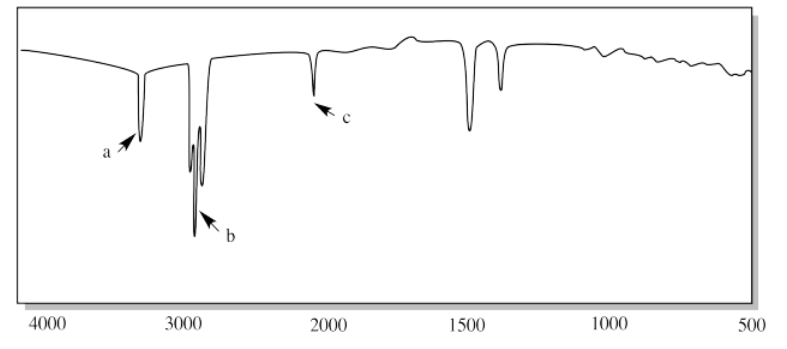
vii.
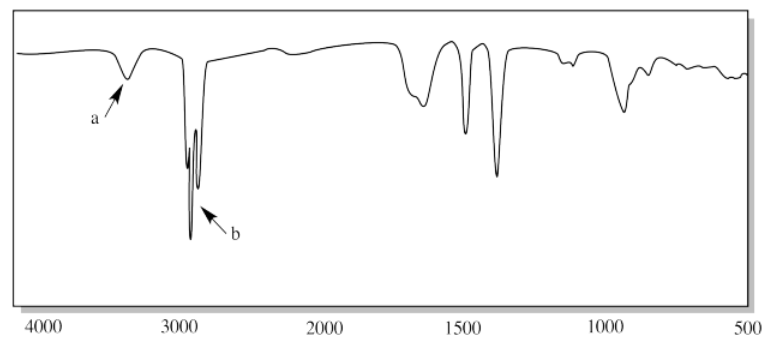
viii.
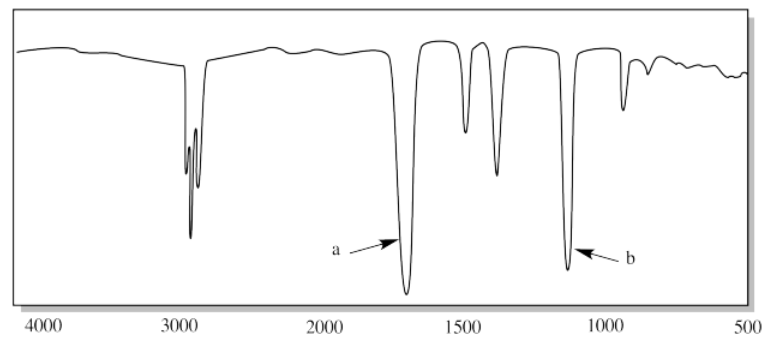
ix.
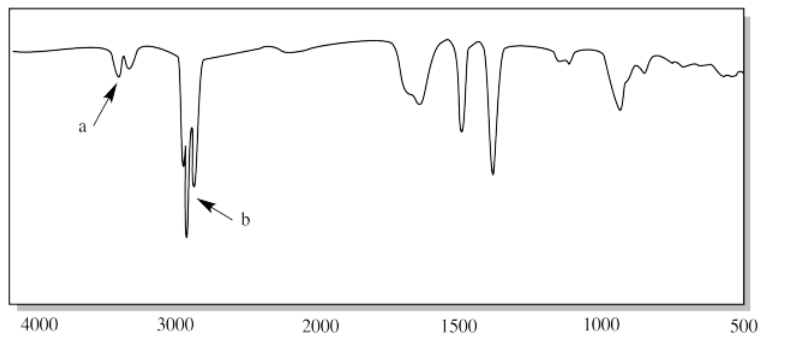
x.
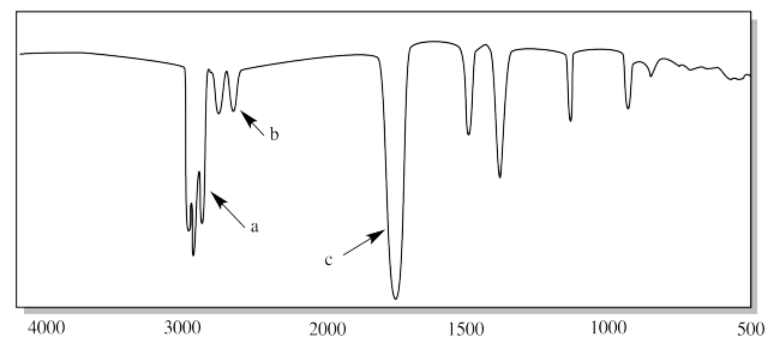
xi.
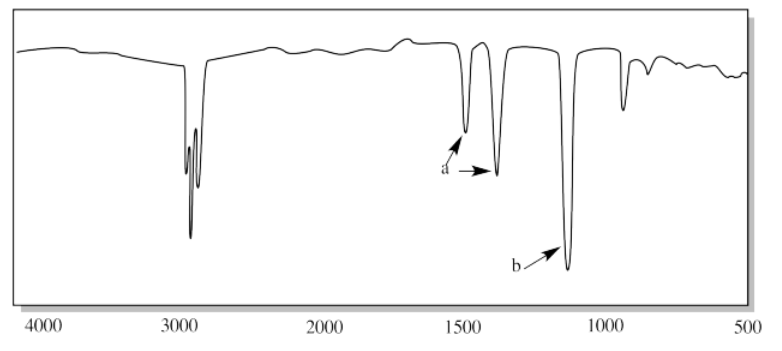
xii.
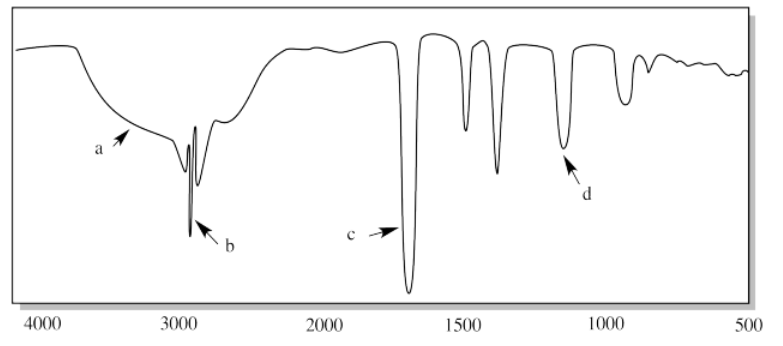
xiii.
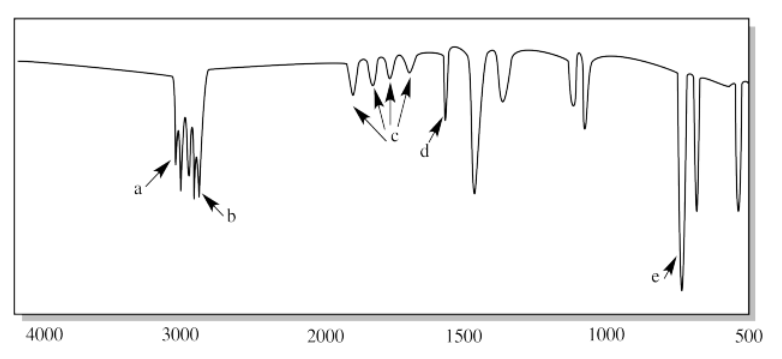
- Answer
-
i. a) O-H b) sp3 C-H c) C-O
ii. a) sp2 C-H b) sp3 C-H c) C=C-H (oop bend)
iii. a) N-H (two of them) b) C=O c) C-N
iv. a) sp3 C-H b) H-C-H (CH2 bend)
v. a) sp3 C-H b) C=O c) H-C-H (CH2 bend)
vi. a) sp C-H b) sp3 C-H c) C=C
vii. a) N-H b) sp3 C-H
viii. a) C=O b) C-O
ix. a) N-H (two of them) b) sp3 C-H
x. a) sp3 C-H b) aldehyde C-H c) C=O
xi. a) H-C-H (CH2 bend) b) C-O
xii. a) O-H (very broad in CO2H) b) sp3 C-H c) C=O d) C-O
xiii. a) sp2 C-H b) sp3 C-H c) aromatic overtones d) C=C
For each of the following hand-drawn cartoons of IR spectra, identify the functional group suggested by the spectrum.
a)
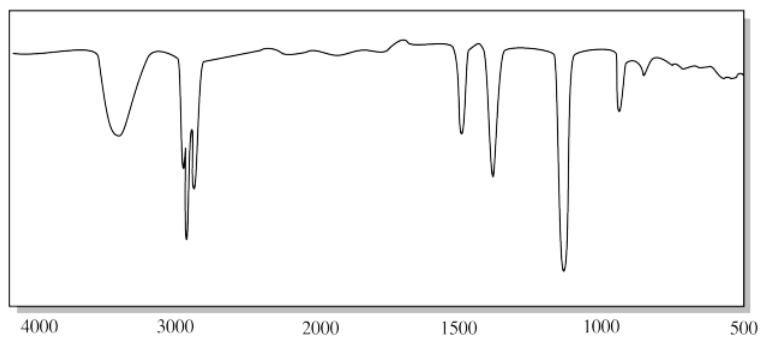
b)
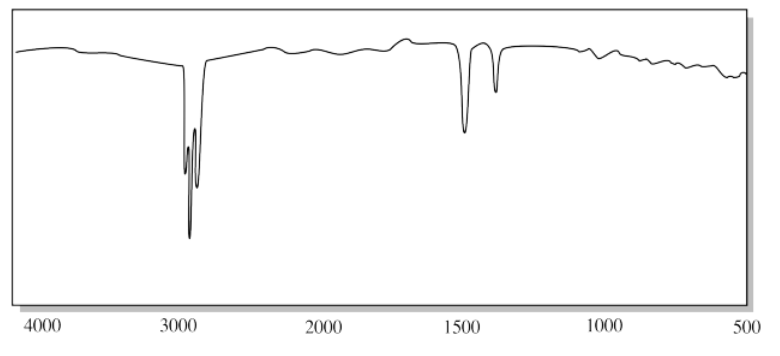
c)
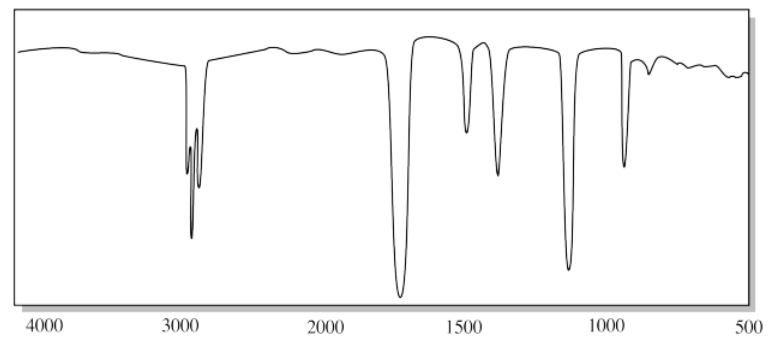
d)
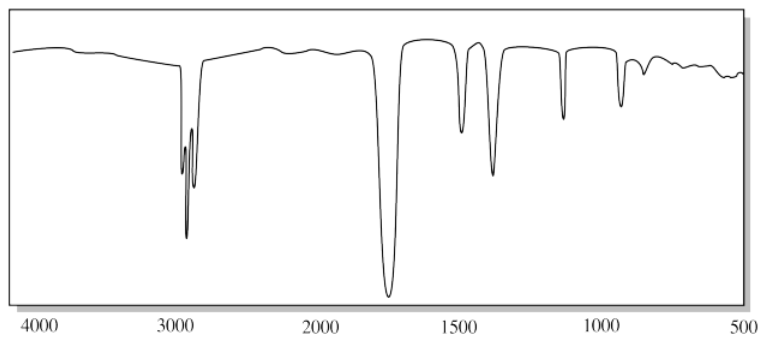
e)
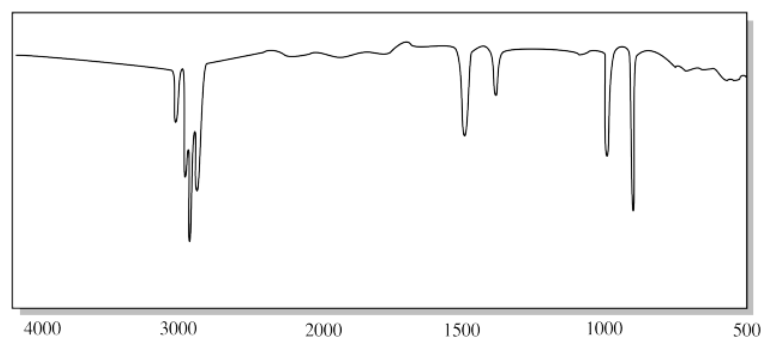
f)
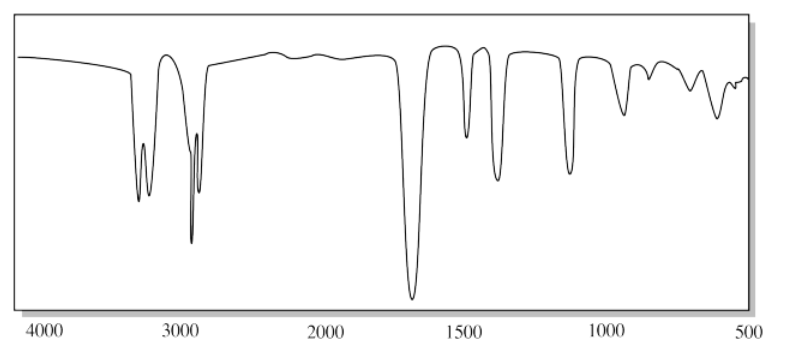
g)
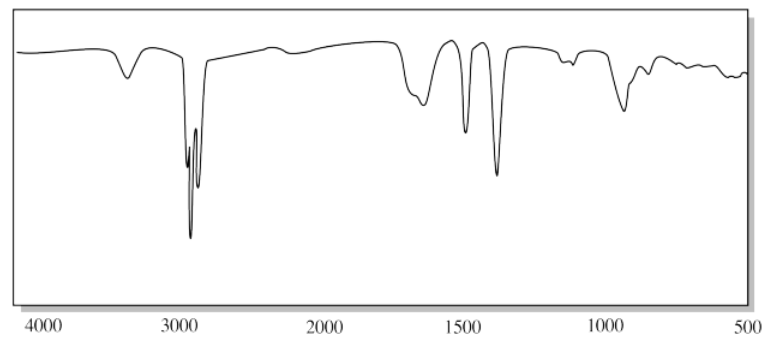
h)
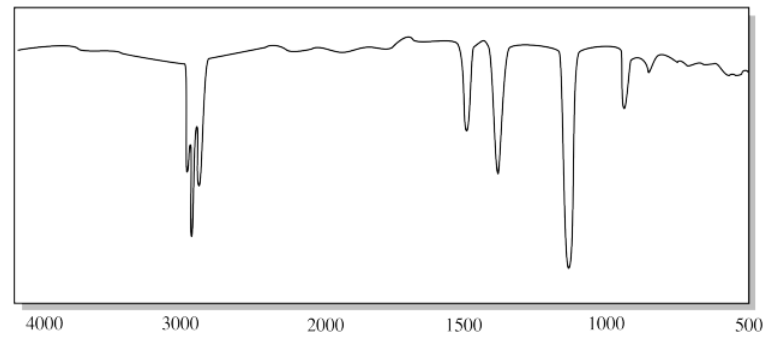
i)
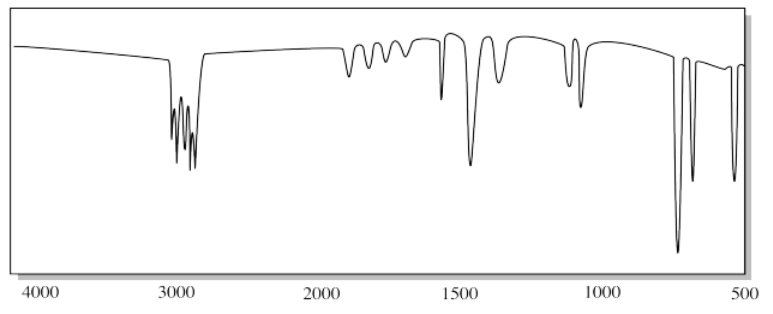
j)
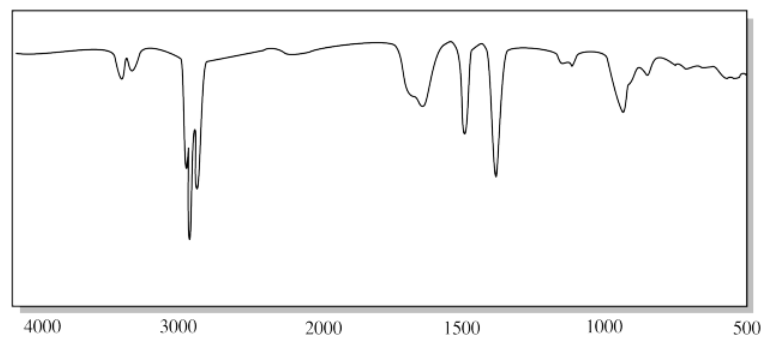
k)
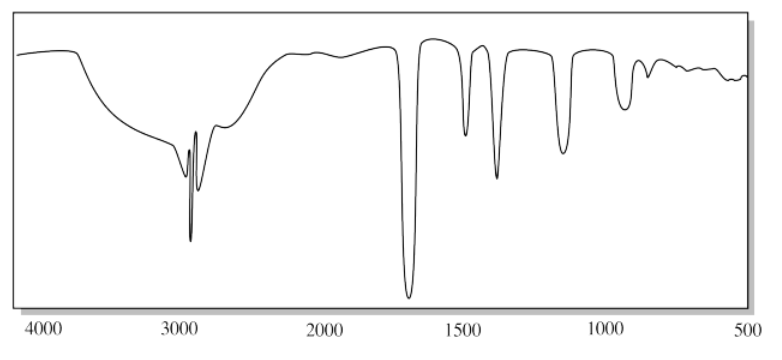
- Answer
-
a) The rounded OH peak near 3300 cm-1 and the strong C-O peak near 1100 cm-1 suggest an alcohol.
b) The sharp CH peaks below 3000 cm-1, the weak CH2 bending modes near 1500 cm-1 and the absence of any other features suggest an alkane.
c) The strong C=O peak near 1700 cm-1 and the strong C-O peak near 1100 cm-1 suggest an ester.
d) The strong C=O peak near 1700 cm-1 and the absence of additional features other than those associated with saturated hydrocarbons suggest a ketone.
e) The CH peak above 3000 cm-1 and the strong oop bending modes below 1000 cm-1 suggest an alkene. The presence of two oop bending peaks may point to a terminal alkene (C=CH2).
f) The sharp N-H peaks near 3200 cm-1 and the strong C=O peak near 1600 cm-1 suggest an amide. the presence of two N-H peaks rather than one points to a primary amide (O=C-NH2).
g) The small, triangular "sharktooth" peak near 3200 cm-1 suggests an amine. With just one N-H peak, this is probably a primary amine (R-NH2).
h) The strong C-O peak near 1100 cm-1 suggests an ether.
i) The C-H peaks above 3000 cm-1 and the oop bending modes below 1000 cm-1 certainly suggest double bonds. The progression of tooth-like "aromatic overtones" between 1600 and 2000 cm-1 strongly indicates a substituted benzene.
j) The small, triangular "sharktooth" peaks near 3200 cm-1 suggest an amine. With two N-H peaks, this is probably a secondary amine (R2NH).
l) The broad, deep OH peak between 3300 cm-1 and 2600 cm-1 and the C=O peak near 1700 cm-1 suggests a carboxylic acid. The O-H peak of a carboxylic acid is often missed; it is moved to lower frequency by hydrogen bonding.
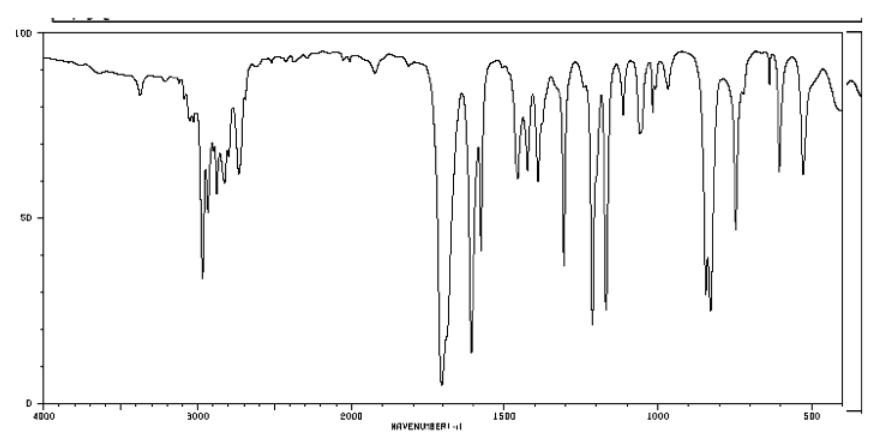
a) Identify at least three important peaks/bonds in each of the following IR spectra.
b) Identify the functional group present in each of these samples. See the Functional Group Section
for help.
c) Draw a possible structure for each of these compounds (there may be many, many correct answers).
problem i)
problem ii)
problem iii)
problem iv)
problem v)
- Answer
-
i)
a)
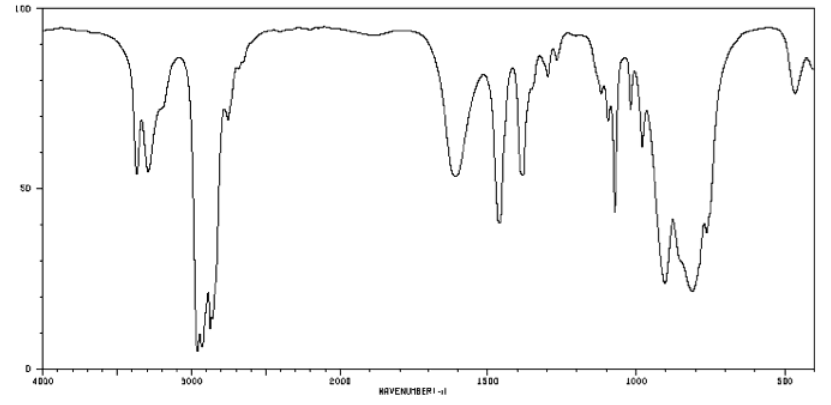
3200 cm-1 (sharp): N-H
2900 cm-1 (sharp): Csp3-H
1600 cm-1 (broad): probably NH2 bending
b) A primary amine.
c)
ii)
a)
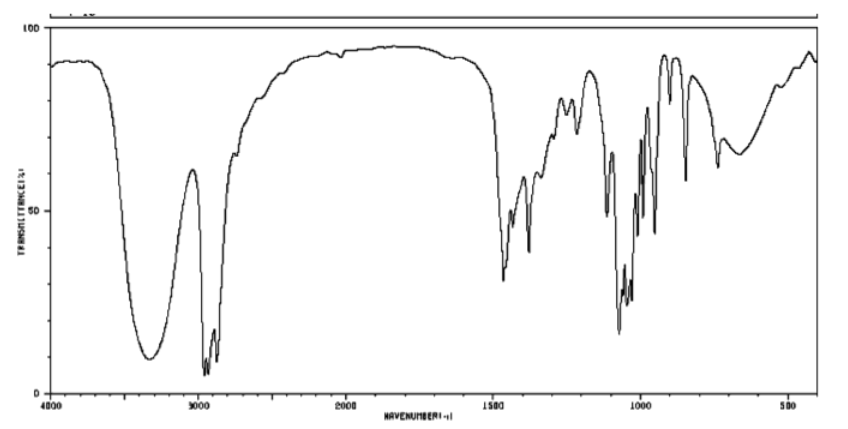
3400 cm-1 (strong, broad): O-H
2900 cm-1 (sharp): Csp3-H
1050 cm-1 (strong): probably C-O
b) Alcohol.
c)
iii)
a)
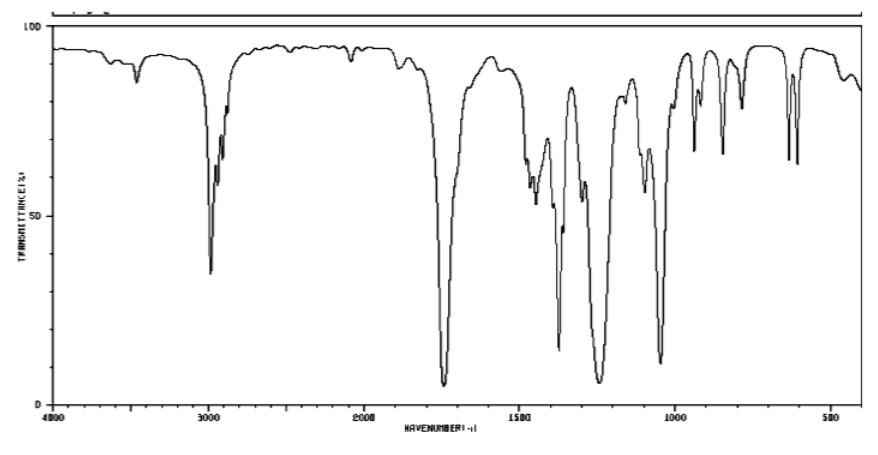
2900 cm-1 (sharp): Csp3-H
1700 cm-1 (strong): C=O
1200 and 1000 cm-1 (strong & medium): C-O
b) Ester
c)
iv)
a)
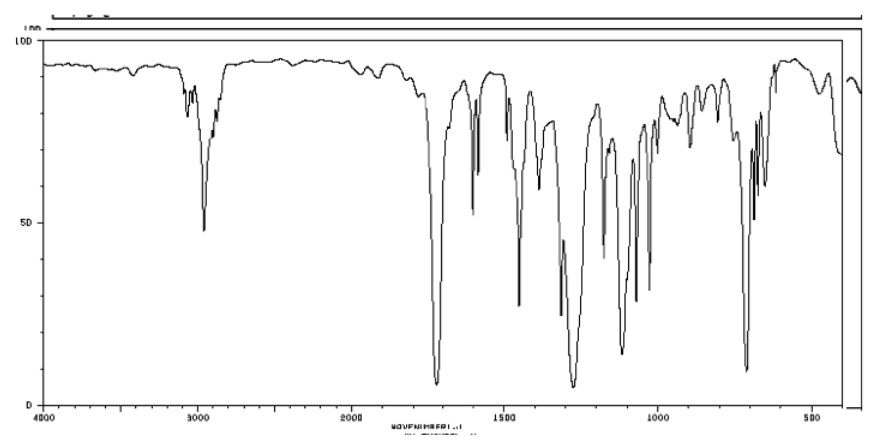
3050 cm-1 (sharp): Csp2-H
1700 cm-1 (strong): C=O
1200 and 1000 cm-1 (strong & medium): C-O
b) Ester; probably contains an aromatic as well
c)
v)
a)
2750 and 2650 cm-1 (medium, sharp): C-H of an aldehyde
1700 cm-1 (strong): C=O
Below 1000 cm-1 (strong & medium): oop bends
b) Aldehyde; probably contains an aromatic as well
Sketch an approximate IR spectrum for each of the following compounds:
The oop bends are sometimes useful in distinguishing substitution pattrens around a benzene ring. Using the spectra of o-, m-, and p-xylene, formulate some guidelines about what the oop bends look like when substituents are one, two or three carbons away on a benzene ring.
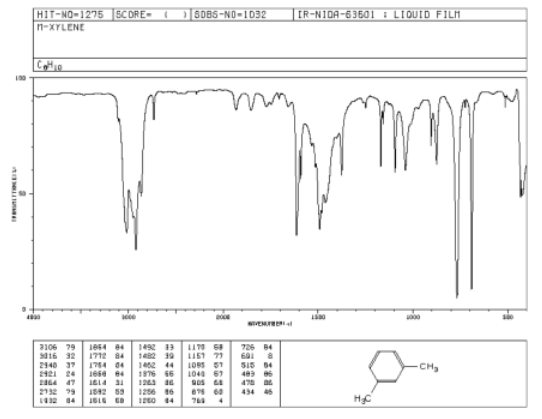
Figure \(\PageIndex{1}\): IR spectrum of m-xylene.
Source: SDBSWeb : http://riodb01.ibase.aist.go.jp/sdbs/ (National Institute of Advanced Industrial Science and Technology of Japan, 14 July 2008)
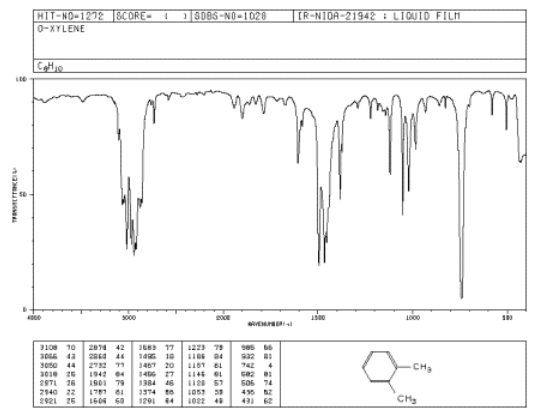
Figure \(\PageIndex{2}\): IR spectrum of o-xylene.
Source: SDBSWeb : http://riodb01.ibase.aist.go.jp/sdbs/ (National Institute of Advanced Industrial Science and Technology of Japan, 14 July 2008)
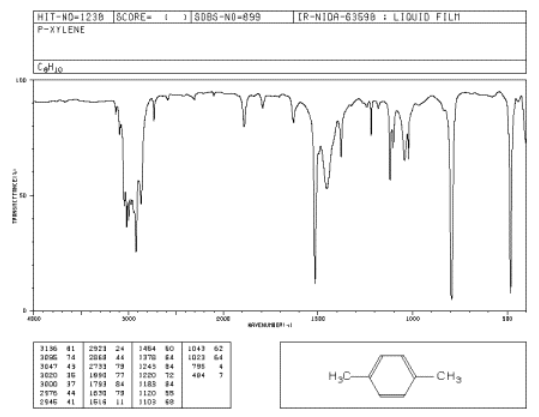
Figure \(\PageIndex{3}\): IR spectrum of p-xylene.
Source: SDBSWeb : http://riodb01.ibase.aist.go.jp/sdbs/ (National Institute of Advanced Industrial Science and Technology of Japan, 14 July 2008


