13.2: Solubility and Molecular Structure
- Page ID
- 6414
- To understand the relationship between solubility and molecular structure.
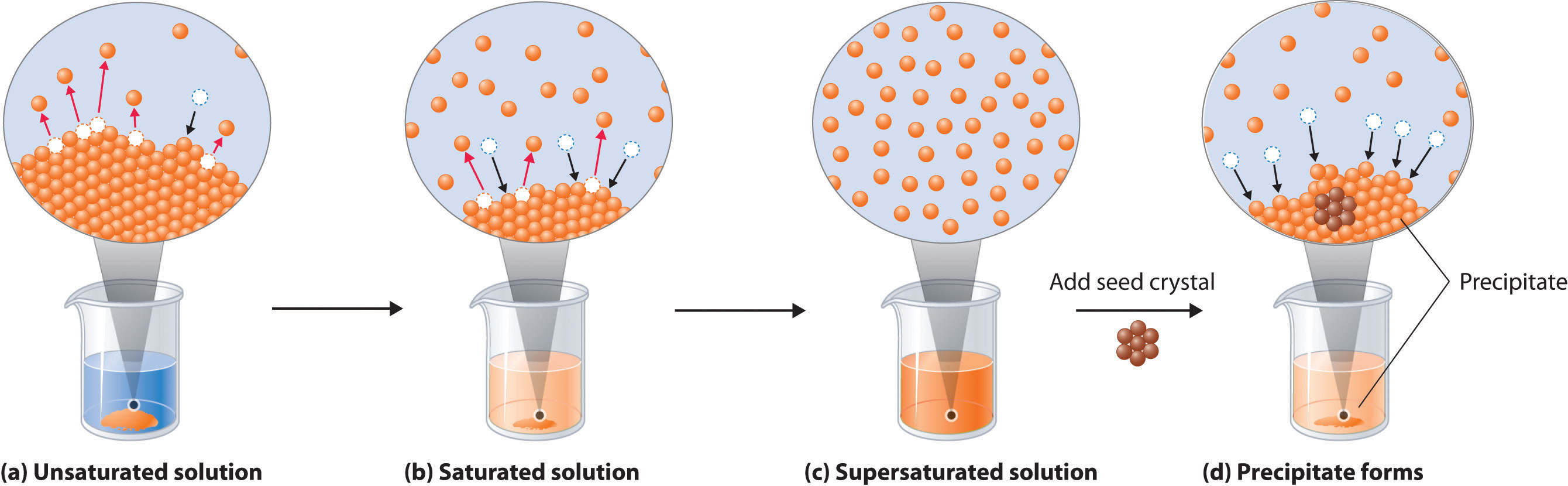
When a solute dissolves, its individual atoms, molecules, or ions interact with the solvent, become solvated, and are able to diffuse independently throughout the solution (part (a) in Figure 13.4). This is not, however, a unidirectional process. If the molecule or ion happens to collide with the surface of a particle of the undissolved solute, it may adhere to the particle in a process called crystallization. Dissolution and crystallization continue as long as excess solid is present, resulting in a dynamic equilibrium analogous to the equilibrium that maintains the vapor pressure of a liquid. We can represent these opposing processes as follows:
\[ \text{solute} + \text{solvent} \ce{<=>[\ce{crystallization}][\ce{dissolution}]} \text{solution} \tag{13.4} \]
Although the terms precipitation and crystallization are both used to describe the separation of solid solute from a solution, crystallization refers to the formation of a solid with a well-defined crystalline structure, whereas precipitation refers to the formation of any solid phase, often one with very small particles.
Contributors and Attributions
- Anonymous


