15.13: Photosynthesis
- Page ID
- 49546
The adjective fossil describes the fossil fuels very aptly, because all of them are derived from the remains of plants or animals which lived on earth millions of years ago. Coal, for example, began as plant matter in prehistoric swamps, where it was able to decompose in the absence of air. Present-day peat bogs are examples of this first stage in coal formation, and in countries such as Ireland dried peat is an important fuel. Over long periods of time, at high temperatures and pressures under the earth’s surface, peat can be transformed into lignite, a brown, soft form of coal. Continued action of geological forces converts lignite into bituminous, or soft coal, and eventually into anthracite, or hard coal. When burned, these latter two types of coal release considerably more heat per unit mass than do lignite or peat.
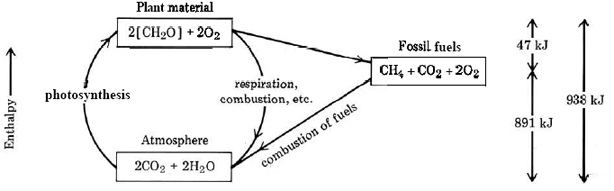
A crucial point to realize about fossil fuels is that the energy we release by burning them came originally from the sun. The plants from which the fuels were derived grew as a result of photosynthesis, the combination of carbon dioxide and water under the influence of sunlight to form organic compounds whose empirical formula is approximately
\[\ce{CO_{2}(g) + H_{2}O(l) \rightarrow [CH_{2}O](s) + O_{2}(g)} \label{1} \]
with \(\Delta H \approx 469 \text{kJ mol}^{-1}\)
Since a number of different substances are formed by photosynthesis, the empirical formula [CH2O] and the ΔH are only approximate.
Photosynthesis is endothermic, and the necessary energy is supplied by the absorption of solar radiant energy. This energy can be released by carrying out the reverse of Equation \ref{1}, a process which is exothermic. When we burn paper, wood, or dried leaves, the heat given off is really a stored form of sunlight. Plants and animals obtain the energy they need to grow or move about from the oxidation of substances produced by photosynthesis. This oxidation process is called respiration.
After millions of years of geological change, the fossil fuels are significantly different in chemical structure from newly photosynthesized plant or animal material. The changes which occur can be approximated by the equation for formation of methane (natural gas):
\[\ce{2[CH_{2}O] \rightarrow CH_{4}(g) + CO_{2}(g)} \nonumber \]
with \(\Delta H \approx –47 \text{kJ mol}^{-1}\).
This reaction is only slightly exothermic, and so very little of the energy captured from sunlight is lost. However, about half the carbon and all the oxygen are lost as carbon dioxide gas, and so a fossil fuel like methane can release more heat per carbon atom (and per gram) than can wood or other organic materials. This is why anthracite and bituminous coals are better fuels than the peat from which they are formed. The enthalpy changes which occur during photosynthesis, respiration, and formation and combustion of fossil fuels are summarized in Figure \(\PageIndex{1}\).