13.10: The Effect of a Change in Pressure
- Page ID
- 49527
As an example of the application of Le Chatelier's principle, consider the effect of tripling the pressure on an equilibrium mixture of NO2 and N2O4:
\[\text{N}_2\text{O}_4(g) \rightleftharpoons \text{2NO}_2(g) \label{1} \]
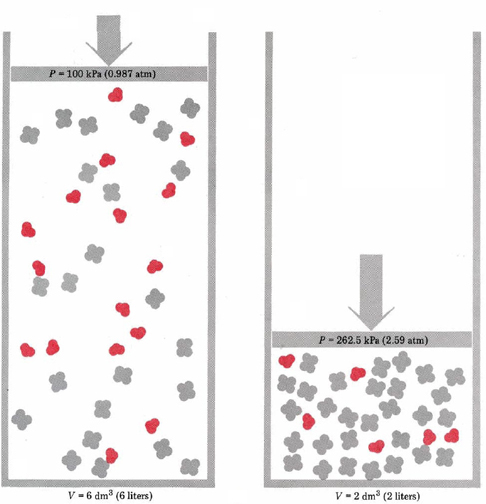
This could be done using the piston and cylinder shown in Figure \(\PageIndex{1}\), in which case tripling the pressure would be expected to reduce the volume of the mixture to one-third its former value. Under the new conditions, however, Le Chatelier’s principle tells us that a new equilibrium will be achieved which counteracts the alteration of pressure. That is, the concentrations of N2O4 and NO2 should change in such a way as to lessen the pressure increase. This can happen if some of the NO2 reacts to form N2O4 because two molecules of NO2 are consumed for every one molecule of N2O4 produced. This reduction in the number of gas molecules will reduce the pressure at the new volume. Thus Le Chatelier’s principle predicts that the reverse of Equation \(\ref{1}\) will occur, producing more N2O4 and using up some NO2. We say that increasing the pressure on the N2O4and NO2 causes the equilibrium to shift to the left. This agrees with the experimental data on this equilibrium, already given in Table 2 in The Equilibrium Constant. Note that the value of the equilibrium constant remains the same, even though the equilibrium shifts.
Notice that the effect just described occurs because the gas volume decreased and the concentrations of NO2 and N2O4 both increased. If we had increased the total pressure on the equilibrium system by pumping in an inert gas such as N2(g), the volume would have remained the same, as would the partial pressures and concentrations of NO2 and N2O4. In such a case no shift in the equilibrium would be expected. Note also the words if possible in the statement of Le Chatelier’s principle. If the equilibrium reaction had not involved a change in the number of molecules in the gas phase, no shift in the concentrations could have made any difference in the pressure. Thus for the reaction
\[\text{2HI}(g) \rightleftharpoons \text{H}_2(g) + \text{I}_2(g) \nonumber \]
changing the pressure by changing the size of the container would have no effect. You can check this for yourself by redoing Example 1 from Calculating the Extent of a Reaction using several different volumes and the same initial amount of HI.
In general, whenever a gaseous equilibrium involves a change in the number of molecules (Δn ≠ 0), increasing the pressure by reducing the volume will shift the equilibrium in the direction of fewer molecules. This applies even if pure liquids or solids are involved in the reaction. An example is the reaction
\[\text{C}(s) + \text{H}_2\text{O}(g) \rightleftharpoons \text{CO}(g) + \text{H}_2(g) \nonumber \]
in which superheated steam is passed over carbon obtained from coal to produce carbon monoxide and hydrogen. Since the volume of solid carbon is negligible compared with the volumes of the gases, we need consider only the latter. Hence Δn = 1 (two gas molecules on the right for every one on the left), and an increase in pressure should favor the reverse reaction. This reaction is an important industrial process, and for the reason we have just outlined, is carried out at a low pressure.
The video below demonstrates what this process looks like, using the reaction of N2O4 and NO2 once again. Here we see visual confirmation of the fact that an increase in the volume of a container causes a shift in the equilibrium based on the number of moles of gas, according to Le Chatelier's principle.


