10.8: Amorphous Materials- Glasses
- Page ID
- 49662
Some liquids become extremely viscous as the temperature falls toward their freezing points, often because they consist of macromolecules. An example is quartz, SiO2, seen .
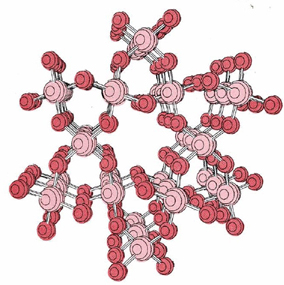

When quartz melts (at 1610°C), a few Si—O bonds break, but most remain intact. The liquid contains large covalently bonded fragments of the original structure and is highly viscous. When the liquid is cooled, the macromolecular fragments cannot readily slide past one another to attain the regular solid structure of quartz. Instead, a collection of interconnected, randomly oriented tetrahedrons of oxygen atoms surrounding silicon atoms is formed, as shown in the figure below. The material having this structure is known as fused silica.
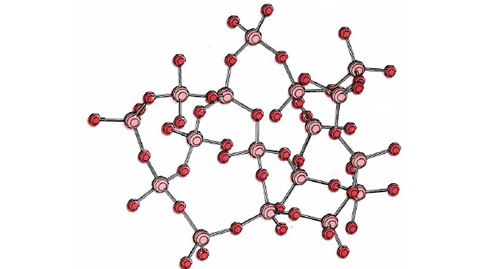
 Fused silica is an example of an amorphous material or glass. It is highly rigid at room temperature, but it does not have the long-range microscopic regularity of a solid crystal lattice. Consequently it cannot be made to cleave along a plane. Instead, like ordinary window glass, it shatters into irregular fragments when struck sharply. (Window glass is primarily silica, but oxides of sodium and calcium are added to lower the melting point.) Since the microscopic structure of a glass is random, like that of a liquid, scientific purists describe glasses as highly viscous liquids, not as solids.
Fused silica is an example of an amorphous material or glass. It is highly rigid at room temperature, but it does not have the long-range microscopic regularity of a solid crystal lattice. Consequently it cannot be made to cleave along a plane. Instead, like ordinary window glass, it shatters into irregular fragments when struck sharply. (Window glass is primarily silica, but oxides of sodium and calcium are added to lower the melting point.) Since the microscopic structure of a glass is random, like that of a liquid, scientific purists describe glasses as highly viscous liquids, not as solids.
To the left is an example of fused silica, which has many industry applications due to its high purity, high melting temperature, and high radiation resistance.


