8.8: Properties of Alkanes
- Page ID
- 49437
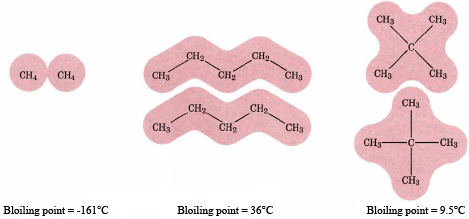
Other things being equal, the more carbon atoms, the higher the boiling point of an alkane. As can be seen from the table of straight chain alkanes, boiling points rise steadily from CH4 which has a boiling point of -162oC to C10H22 which has a boiling point of 174oC as each additional CH2 group is added to the chain.
The reason for this rise is an increase in the intermolecular forces. As shown in the following figure, larger molecules can make close contact with each other over a much larger surface area than can smaller molecules. The total force exerted between the two is thus greater. In the same way we can also explain why branched-chain hydrocarbons boil at lower temperatures than straight-chain compounds. The branched molecules are more compact and provide less area over which intermolecular forces can act.
The alkanes are rather unreactive and do not combine readily with other substances. When heated sufficiently, however, they burn in air, a process known as combustion:
\[\text{CH}_{\text{4}}(g) + \text{ 2O}_{\text{2}}(g)\rightarrow \text{ CO}_{\text{2}}(g) + \text{ 2H}_{\text{2}}\text{O}(g) \nonumber \] ΔHm = –890.4 kJ mol–1
The following video showcases this combustion reaction of methane with oxygen:
Two balloons, one filled with methane, and one filled with a 1:1 by volume mixture of methane and oxygen are ignited. The video also shows these reactions in the dark, where it is easier to see the difference. The oxygen/methane mixture balloon explodes much more violently. Looking at the equation above, it is easy to understand why. The mixture in that balloon is much closer to the stoichiometry of the reaction than the pure methane balloon, which must react with oxygen in the atmosphere which it is not mixed with. Note that the major products of this reaction are water, CO2 and heat. Combustion releases large quantities of heat; not surprisingly, the most important use of alkanes is as fuels. Natural gas is mainly (approximately 85 percent) methane.
Propane or liquefied petroleum gas sold in tanks for portable use is usually a mixture of both propane and butane. Though both are gases at ordinary temperature and pressure, they liquefy under pressure in the tank. Gasoline is a more complex mixture of alkanes having 5 to 12 carbon atoms. Better-quality gasoline usually contains a higher percentage of branch-chain alkanes than the regular grade. Kerosene contains C10 to C16 alkanes, while heating oil usually involves the C12 to C18 range. Even longer-chain alkanes are found in lubricating oil, C15 to C25 and paraffin wax (used for candles and waxed paper), C23 to C29.
The variation of boiling point with chain length in the alkanes provides a simple method for partially separating them from each other in petroleum. When petroleum is heated, the shorter-chain compounds begin to boil off initially. These can be collected by cooling the vapor until it recondenses to a liquid. As boiling proceeds, the temperature rises and longer and longer-chain compounds boil off. Finally only the very long chain compounds are left. Such a process is called fractional distillation and is discussed more in depth in the section on distillation. Fractional distillation plays an important role in petroleum refining, which is a series of physical and chemical processes by which the most valuable and useful components are obtained from natural crude oil.
The alkanes are typical examples of nonpolar compounds. The electronegativities of carbon and hydrogen are 2.5 and 2.1, respectively, and so the C—H bond has almost no polarity. Consequently, even the most unsymmetrical hydrocarbon molecules have very small dipole moments. We can therefore take the boiling-point behavior of the alkanes to be representative of other nonpolar substances. Simple molecules are gases, while medium-sized molecules are liquids, and only quite large molecules are solids.
Chirality
Alkanes have carbons with four single bonds. In most examples you have seen, these are all to either hydrogen or to other carbon atoms. Later, we will look at molecules where carbon is bonded to other elements such as oxygen or nitrogen. However, even when carbon only bonds to hydrogen and carbon, we still see a type of stereoisomerism that is different from cis/trans isomerism. Take a look at
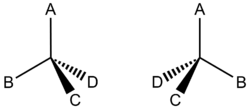
A simple example of two things which are stereoisomers is our hands. No matter what you do, you cannot put one hand on top of the other and have both of the palms facing down and the thumbs pointing in the same direction. The same is true for the molecules in Figure 1. There is no way to rotate one molecule so that A points up, and all of the other species point in similar directions to the other molecule. To obtain an optical isomer of a chiral carbon, the stereocenter must be inverted. This is much like reflection through a mirror. If you hold a right hand up to a mirror and compare it to your left hand (not reflected through a mirror), you will find that the mirror image of your right hand matches your left hand!
In order to find chiral carbons in a simple alkane, find any carbon which has four different things attached to it. For instance, in the following molecule, see how there is a hydrogen, a three-carbon chain (propyl group), a two-carbon chain (ethyl group), and a one-carbon chain (methyl group) attached to the central carbon.
| Rotate this molecule (3-methylhexane) to view how the 3rd carbon along the longest chain is chiral. |
Find all chiral carbons in the following molecules.

Solution. First, we will name the molecule according to IUPAC guidelines. The molecule on the left is called 2,5-dimethylheptane, the numbering starts so as to get the lowest overall numbers and therefore begins with carbon 1 on the far right. This molecule has two possibilities. Carbons 2 and 5, as all other carbons have at least two hydrogens and therefore cannot be chiral. Carbon 2 has a hydrogen, the long chain, and then two methyl groups and thus is not chiral. Carbon 5 has a hydrogen, a methyl, an ethyl and then a long carbon chain and thus is indeed chiral.
The second molecule is named 3,4,5-trimethylheptane and is symmetrical, so the numbering is the same from either side. Due to this symmetry, the center carbon (carbon 4) cannot be chiral (the two long chains are identical). Thus only carbons 3 and 5 are chiral. Both molecules are shown again below with their chiral carbons - or stereocenters - indicated by a black dot.