7.9: Polarizability
- Page ID
- 49308
In other sections, chemical bonds are divided into two classes: covalent bonds, in which electrons are shared between atomic nuclei, and ionic bonds, in which electrons are transferred from one atom to the other. However, a sharp distinction between these two classes cannot be made. Unless both nuclei are the same (as in H2), an electron pair is never shared equally by both nuclei. There is thus some degree of electron transfer as well as electron sharing in most covalent bonds. On the other hand there is never a complete transfer of an electron from one nucleus to another. The first nucleus always maintains some slight residual control over the transferred electron.
The effects on a bond due to different electronegativity values can be observed by studying carefully the lithium hydride ion pair discussed in "Ionic Bonding". Figure \(\PageIndex{1}\) shows a dot-density diagram of the 1s2 electron cloud of the hydride ion, H–, as well as the two nuclei. For the sake of clarity the two electrons around the Li nucleus have been omitted. Were it not for the presence of the Li+ ion on the right-hand side of the diagram, we could expect a spherical (or, in the two dimensions shown, a circular) distribution of electron density around the H nucleus.
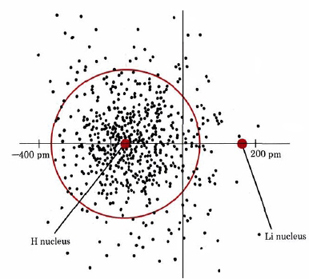
As can be seen by comparing the density of dots to the left of the colored circle in Figure \(\PageIndex{1}\) with that to the right, the actual distribution is not exactly circular. Instead the electron cloud is distorted by the attraction of the Li+ ion so that some of the H– 1s2 electron density is pulled into the bonding region between the Li and H nuclei. This contributes partial covalent character to the bond.
Distortion of an electron cloud, as described in the previous paragraph, is called polarization. The tendency of an electron cloud to be distorted from its normal shape is referred to as its polarizability. The polarizability of an ion (or an atom) depends largely on how diffuse or spread out its electron cloud is.
For example, most positive ions have relatively small radii, and their electrons are held rather tightly by the excess of protons in the nucleus. Thus their polarizabilities are usually small. Only quite large positive ions such as Cs+ are significantly polarizable. On the other hand, negative ions have excess electrons, large radii, and diffuse electron clouds which can be polarized easily. Thus negative ions, especially large ones, have high polarizabilities. Small, highly charged positive ions can distort them quite extensively.
The slight shift in the electron cloud of H– shown in Figure 1 can be confirmed experimentally. An ion pair like LiH has a negative end (H–) and a positive end (Li+). That is, it has two electrical “poles,” like the north and south magnetic poles of a magnet. The ion pair is therefore an electrical dipole (literally “two poles“), and a quantity known as its dipole moment may be determined from experimental measurements. The dipole moment μ is proportional to the size of the separated electrical charges Q and to the distance r between them:\[μ = Qr \tag{1} \]
In the LiH ion pair the two nuclei are known to be separated by a distance of 159.5 pm. If the bond were completely ionic, there would be a net charge of –1.6021 × 10–19 C (the electronic charge) centered on the H nucleus and a charge of +1.6021 × 10–19 C centered on the Li nucleus:
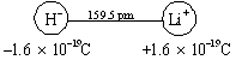
The dipole moment would then be given by
\(μ = Qr = 1.6021 \times 10^{-19} \text{C} \times 159.5 \times 10^{-12} \text{m} = 2.555 \times 10^{-29} \text{C m}\)
The measured value of the dipole moment for the LiH ion pair is only about 77 percent of this value, namely, 1.963 × 10–29 C m. This can only be because the negative charge is not centered on the H nucleus but shifted somewhat toward the Li+ nucleus. This shift brings the opposite charges closer together, and the experimental dipole moment is smaller than would be expected.
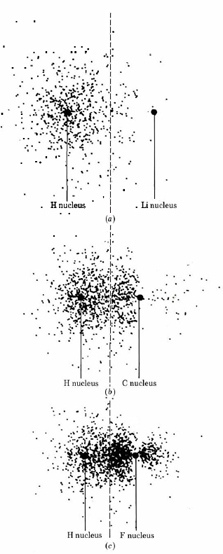
If we increase the degree of polarization of an ionic bond, a bond which is more covalent than ionic is eventually obtained. This is illustrated in Figure \(\PageIndex{2}\). Three bonds involving hydrogen are shown, and the diagrams are arranged so that the midpoint of each bond lies on the same vertical (dashed) line. We have already discussed the bond between hydrogen and lithium, in which most of the electron density is associated with hydrogen. By comparison, electron density in the bond between hydrogen and carbon is much more evenly distributed—the bond certainly appears to be covalent. The third bond involves fluorine, which is three places farther to the right along the second row of the periodic table than carbon. In the H―F bond, electron density has been distorted away from hydrogen even more. Thus as we move from lithium with a nuclear charge of +3, through carbon with a nuclear charge of +6, to fluorine with a nuclear charge of +9, there is a continual shift in electron density away from hydrogen. The original H– ion is polarized to the point where much of its electron density has been removed, and it begins to look more like an H+ ion.