6.8: Electron Affinities
- Page ID
- 49351
Electron affinities are more difficult to measure experimentally than are ionization energies, and far fewer values are available. The relationship of the periodic table with those electron affinities that have been measured or estimated from calculations can be seen on the table of ionization energies and electron affinities (in the red), seen below.
Table \(\PageIndex{1}\) Ionization Energies and Electron Affinities. The electron affinities are the red values above the atomic symbol.
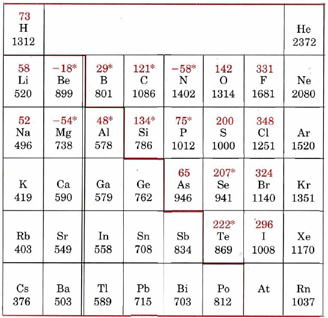
* Electron affinities marked with an asterisk (*) have been obtained from theoretical calculations rather than experimental measurements. The heavy colored line separates metals (ionization energy usually below about 800 kJ mol–1) from nonmetals.
It is not easy to discern many obvious regularities in this table, especially since some of the electron-affinity values quoted are negative, indicating that energy is sometimes required to force an extra electron onto an atom. Nevertheless, it is quite obvious which of the periodic groups correspond to the highest electron affinities. All the halogens have values of about 300 kJ mol–1 while the group VI nonmetals have somewhat lower values, in the region of 200 kJ mol–1 or less. The high electron affinities of the halogens are a result of their having an almost complete outer shell of electrons.
The element fluorine, for example, has the structure 1s22s22p5, in which one of the 2p orbitals contains but one electron. If an extra electron (in the red in both diagrams below) is added to this atom to form a fluoride ion, the electron can pair with the electron in the half-filled 2p orbital (as seen in the orbital diagram below).
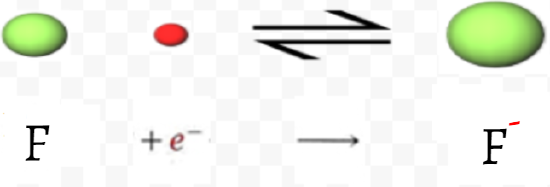
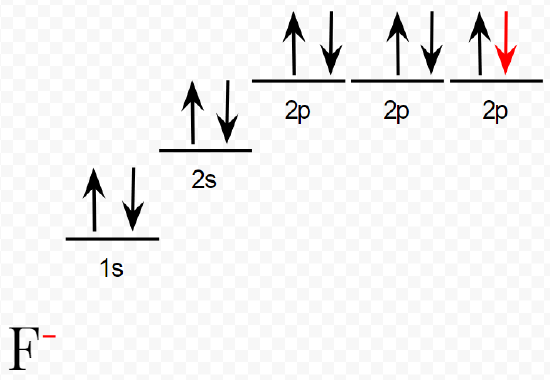
The added electron will be shielded from the nucleus by the 1s electrons, but the 2s and 2p electrons are in the same shell and will shield it rather poorly. There will thus be quite a large effective nuclear charge (a rough estimate is +5) attracting the added electron. Because of this overall attraction, energy will be released when the electron is captured by the fluorine atom. Similar reasoning also explains why oxygen also has a high electron affinity. Here, though, the nuclear charge is smaller, and the attraction for the added electron distinctly less.


