5.17: Electron Configurations and the Periodic Table
- Page ID
- 49338
The commonly used long form of the periodic table is designed to emphasize electron configurations. Since it is the outermost (valence) electrons which are primarily involved in chemical interactions between atoms, the last electron added to an atom in the building-up process is of far more interest to a chemist than the first. This last electron is called the distinguishing electron because it distinguishes an atom from the one immediately preceding it in the periodic table. The type of subshell (s, p, d, f)into which the distinguishing electron is placed is very closely related to the chemical behavior of an element and gives rise to the classification shown by the color-coding on the periodic table seen here. The representative elements are those in which the distinguishing electron enter ans or p subshell. Most of the elements whose chemistry and valence we have discussed so far fall into this category. Many of the chemical properties of the representative elements can be explained on the basis of Lewis diagrams. That is, the valences of the representative elements may be predicted on the basis of the number of valence electrons they have, or from the number of electrons that would have to be added in order to attain the same electron configuration as an atom of a noble gas. For representative elements the number of valence electrons is the same as the periodic group number, and the number needed to match the next noble-gas configuration is 8 minus the group number. This agrees with the valence rules derived from the periodic table, and results in formulas for chlorides of the first dozen elements that show the periodic variation of valence.
| Element | Atomic Weight | Hydrogen Compounds | Oxygen Compounds | Chlorine Compounds |
|---|---|---|---|---|
| Hydrogen | 1.01 | H2 | H2O, H2O2 | HCl |
| Helium | 4.00 | None formed | None formed | None formed |
| Lithium | 6.94 | LiH | Li2O, Li2O2 | LiCl |
| Beryllium | 9.01 | BeH2 | BeO | BeCl2 |
| Boron | 10.81 | B2H6 | B2O3 | BCl3 |
| Carbon | 12.01 | CH4, C2H6, C3H8 | CO2, CO, C2O3 | CCl4, C2Cl6 |
| Nitrogen | 14.01 | NH3, N2H4, HN3 | N2O, NO, NO2, N2O5 | NCl3 |
| Oxygen | 16.00 | H2O, H2O2 | O2, O3 | <Cl2O, ClO2, Cl2O7 |
| Fluorine | 19.00 | HF | OF2, O2F2 | ClF, ClF3, ClF5 |
| Neon | 20.18 | None formed | None formed | None formed |
| Sodium | 22.99 | NaH | Na2O, Na2O2 | NaCl |
| Magnesium | 24.31 | MgH2 | MgO | MgCl2 |
The first three horizontal rows or periods in the modern periodic table consist entirely of representative elements. In the first period the distinguishing electrons for H and He are in the 1s subshell. Across the second period Li and Be have distinguishing electrons in the 2s subshell, and electrons are being added to the 2p subshell in the atoms from B to Ne. In the third period the 3s subshell is filling for Na and Mg, and therefore Al, Si, P, S, Cl, and Ar. As a general rule, in the case of the representative elements, the distinguishing electron will be in an ns or np subshell. The value of n, the principal quantum number for the distinguishing electron, can be quickly determined by counting down from the top of the periodic table. For example, iodine is a representative element in the fifth period. Therefore the distinguishing electron must occupy either the 5s or 5p subshell. Since I is on the right side of the table, 5p is the correct choice.
When the principal quantum number is three or more, d-type subshells are also possible. The transition elements or transition metals are those elements whose distinguishing electron is found in a d orbital. The first examples of transition metals (Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn) are found in the fourth period even though the distinguishing electron in each case is a 3d electron and belongs to the third shell. This hiatus results, as we have already seen, because the 4s is lower in energy than the 3d. The 4s orbital thus starts to fill up, beginning the fourth period before any of the 3d orbitals can become occupied.
Figure \(\PageIndex{1}\) compares the probability distributions of a 4s and a 3d electron in a V atom. Although the 4s electron cloud lies farther from the nucleus on average than does the 3d cloud, a small portion of the 4s electron density is found very close to the nucleus where it is hardly shielded from the total nuclear charge of +23. It is the very strong attractive force of this small fraction of the total 4s electron density that lowers the energy of the 4s electron below that of the 3d.
_and_4s_(color)_Electron_Clouds.jpg?revision=1)
The fact that the 4s electron cloud is more extensive than the 3d has an important influence on the chemistry of the transition elements. When an atom such as V (Figure \(\PageIndex{1}\) ) interacts with another atom, it is the 4s electrons extending farthest from the nucleus which first contact the other atom. Thus the 4s electrons are often more significant than the 3d in determining valence and the formulas of compounds. The 3d electrons are “buried” under the surfaces of the atoms of the transition metals. Adding one more 3d electron has considerably less effect on their chemical properties than adding one more 3s or 3p electron did in the case of the representative elements. Hence there is a slow but steady transition in properties from one transition element to another. Notice, for example, that except for Sc, all of the transition metals form chlorides, MCl2, where the metal has a valence of 2; examples are TiCl2, VCl2, CrCl2, and so on. This can be seen in the table found at the top of this page. The valence of 2 corresponds with the two 4s valence electrons.
Each of the transition metals also exhibits other valences where one or more of the 3d electrons are also involved. For example, in some compounds V (vanadium) has a valence of 2 (VO, VCl2) in others it has a valence of 3 (V2O3, VCl3), in still others it has a valence of 4 (VO2, VCl4), and in at least one case (V2O5) it has a valence of 5. The chemistry of the transition metals is more complicated and a wider variety of formulas for transition-metal compounds is possible because of this variable valence. In some cases electrons in the d subshells act as valence electrons, while in other cases they do not. Although the 3d electron clouds do not extend farther from the nucleus than 3s and 3p (and hence do not constitute another shell as the 4s electrons do), they are thoroughly shielded from the nuclear charge and thus often act as valence electrons. This Jekyll and Hyde behavior of 3d electrons makes life more complicated (and often far more interesting) for chemists who study the transition elements.
| Z | Element | Configuration |
|---|---|---|
| 1 | H | 1s 1 |
| 2 | He | 1s 2 |
| 3 | Li | [He] 2s 1 |
| 4 | Be | [He] 2s 2 |
| 5 | B | [He] 2s 2 2p1 |
| 6 | C | [He] 2s 2 2p2 |
| 7 | N | [He] 2s 2 2p3 |
| 8 | 0 | [He] 2s 2 2p4 |
| 9 | F | [He] 2s 2 2p5 |
| 10 | Ne | [He] 2s 2 2p6 |
| 11 | Na | [Ne] 3s 1 |
| 12 | Mg | [Ne] 3s 2 |
| 13 | Al | [Ne] 3s 2 3p1 |
| 14 | Si | [Ne]3s 2 3p2 |
| 15 | P | [Ne] 3s 2 3p3 |
| 16 | S | [Ne] 3s 2 3p4 |
| 17 | Cl | [Ne] 3s 2 3p5 |
| 18 | Ar | [Ne] 3s 2 3p6 |
| 19 | K | [Ar] 4s 1 |
| 20 | Ca | [Ar] 4s 2 |
| 21 | Sc | [Ar] 3d 1 4s 2 |
| 22 | Ti | [Ar] 3d 2 4s 2 |
| 23 | V | [Ar] 3d 3 4s 2 |
| 24 | Cr | [Ar] 3d 5 4s 1 |
| 25 | Mn | [Ar] 3d 5 4s 2 |
| 26 | Fe | [Ar] 3d 6 4s 2 |
| 27 | Co | [Ar] 3d 7 4s 2 |
| 28 | Ni | [Ar] 3d 8 4s 2 |
| 29 | Cu | [Ar] 3d 10 4s 1 |
| 30 | Zn | [Ar] 3d 10 4s 2 |
| 31 | Ga | [Ar] 3d 10 4s 2 4p 1 |
| 32 | Ge | [Ar] 3d 10 4s 2 4p 2 |
| 33 | As | [Ar] 3d 10 4s 2 4p 3 |
| 34 | Se | [Ar] 3d 10 4s 2 4p 4 |
| 35 | Br | [Ar] 3d 10 4s 2 4p 5 |
| 36 | Kr | [Ar] 3d 10 4s 2 4p 6 |
| 37 | Rb | [Kr] 5s 1 |
| 38 | Sr | [Kr] 5s 2 |
| 39 | Y | [Kr] 4d 1 5s 2 |
| 40 | Zr | [Kr] 4d 2 5s 2 |
| 41 | Nb | [Kr] 4d 4 5s 1 |
| 42 | Mo | [Kr] 4d 5 5s 1 |
| 43 | Tc | [Kr] 4d 5 5s 2 |
| 44 | Ru | [Kr] 4d 7 5s 1 |
| 45 | Rh | [Kr] 4d 8 5s 1 |
| 46 | Pd | [Kr] 4d 10 |
| 47 | Ag | [Kr] 4d 10 5s 1 |
| 48 | Cd | [Kr] 4d 10 5s 2 |
| 49 | In | [Kr] 4d 10 5s 2 5p 1 |
| 50 | Sn | [Kr] 4d 10 5s 2 5p 2 |
| 51 | Sb | [Kr] 4d 10 5s 2 5p 3 |
| 52 | Te | [Kr] 4d 10 5s 2 5p 4 |
| 53 | I | [Kr] 4d 10 5s 2 5p 5 |
| 54 | Xe | [Kr] 4d 10 5s 2 5p 6 |
| 55 | Cs | [Xe] 6s 1 |
| 56 | Ba | [Xe] 6s 2 |
| 57 | La | [Xe] 5d 1 6s 2 |
| 58 | Ce | [Xe] 4f 1 5d 1 6s 2 |
| 59 | Pr | [Xe] 4f 3 6s 2 |
| 60 | Nd | [Xe] 4f 4 6s 2 |
| 61 | Pm | [Xe] 4f 5 6s 2 |
| 62 | Sm | [Xe] 4f 6 6s 2 |
| 63 | Eu | [Xe] 4f 7 6s 2 |
| 64 | Gd | [Xe] 4f 7 5d 1 6s 2 |
| 65 | Tb | [Xe] 4f 9 6s 2 |
| 66 | Dy | [Xe] 4f 10 6s 2 |
| 67 | Ho | [Xe] 4f 11 6s 2 |
| 68 | Er | [Xe] 4f 12 6s 2 |
| 69 | Tm | [Xe] 4f 13 6s 2 |
| 70 | Yb | [Xe] 4f 14 6s 2 |
| 71 | Lu | [Xe] 4f 14 5d 1 6s 2 |
| 72 | Hf | [Xe] 4f 14 5d 2 6s 2 |
| 73 | Ta | [Xe] 4f 14 5d 3 6s 2 |
| 74 | W | [Xe] 4f 14 5d 4 6s 2 |
| 75 | Re | [Xe] 4f 14 5d 5 6s 2 |
| 76 | 0s | [Xe] 4f 14 5d 6 6s 2 |
| 77 | Ir | [Xe] 4f 14 5d 7 6s 2 |
| 78 | Pt | [Xe] 4f 14 5d 9 6s 1 |
| 79 | Au | [Xe] 4f 14 5d 10 6s 1 |
| 80 | Hg | [Xe] 4f 14 5d 10 6s 2 |
| 81 | Tl | [Xe] 4f 14 5d 10 6s 2 6p1 |
| 82 | Pb | [Xe] 4f 14 5d 10 6s 2 6p2 |
| 83 | Bi | [Xe] 4f 14 5d 10 6s 2 6p 3 |
| 84 | Po | [Xe] 4f 14 5d 10 6s 2 6p 4 |
| 85 | At | [Xe] 4f 14 5d 10 6s 2 6p 5 |
| 86 | Rn | [Xe] 4f 14 5d 10 6s 2 6p 6 |
| 87 | Fr | [Rn] 7s 1 |
| 88 | Ra | [Rn] 7s 2 |
| 89 | Ac | [Rn] 6d 1 7s 2 |
| 90 | Th | [Rn] 6d 2 7s 2 |
| 91 | Pa | [Rn] 5f 2 6d 1 7s 2 |
| 92 | U | [Rn] 5f 3 6d 1 7s 2 |
| 93 | Np | [Rn] 5f 4 6d 1 7s 2 |
| 94 | Pu | [Rn] 5f 6 7s 2 |
| 95 | Am | [Rn] 5f 7 7s 2 |
| 96 | Cm | [Rn] 5f 7 6d 1 s 2 |
| 97 | Bk | [Rn] 5f 9 s 2 |
| 98 | Cf | [Rn] 5f 10 s 2 |
| 99 | Es | [Rn] 5f 11 s 2 |
| 100 | Fm | [Rn] 5f 12 s 2 |
| 101 | Md | [Rn] 5f 13 s 2 |
| 102 | No | [Rn] 5f 14 s 2 |
| 103 | Lr | [Rn] 5f 14 6d 1 s 2 |
| 104 | Rf | [Rn] 5f 14 6d 2 s 2 |
The third major category of elements arises when the distinguishing electron occupies an f subshell. The first example occurs in the case of the lanthanoids (elements having atomic numbers between 57 and 71).The lanthanoids have the general electron configuration
-
- [Kr]4d104f i5s25p65d0 or 16s2
where i is a number between 0 and 14. Thus in the building-up process for the lanthanoids, electrons are being added to a subshell (4f) whose principal quantum number is two less than that of the outermost orbital (6s). Addition of another electron to an inner shell buried as deeply as the 4f has little or no effect on the chemical properties of these elements. All are quite similar to lanthanum (La) and might fit into exactly the same space in the periodic table as La. The lanthanoid elements are so similar to one another that special techniques are required to separate them. As a result, even approximately pure samples of most of them were not prepared until the 1870s. Following the element actinium (Ac) is a series of atoms in which the 5f subshell is filling. The actinoids are somewhat less similar to Ac than the lanthanoids are to La because some exceptions to the usual order of filling orbitals occur in the case of Th, Pa, and U (Table \(\PageIndex{1}\) ).
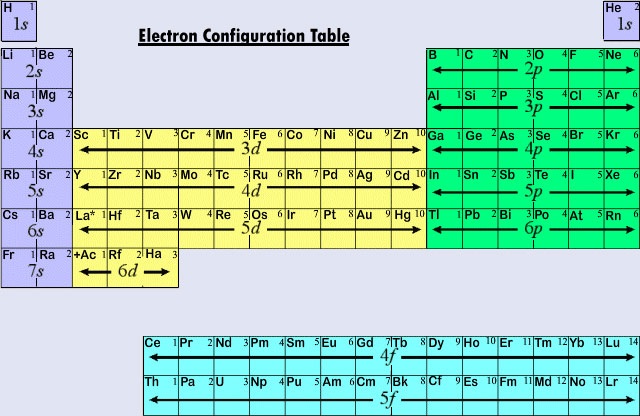
Because the lanthanoids and most of the actinoids behave chemically as if they should fit in group IIIB of the periodic table (where Lu and Lr are found), both groups are separated from the rest of the table and placed together in a block below. Taken together, the lanthanoids and actinoids are called inner transition elements because the f subshells being filled lie so deep within the remaining electronic structure of their atoms.
Figure \(\PageIndex{2}\) summarizes the type of subshell in which the distinguishing electron is to be found for atoms of elements in various regions of the periodic table. This summary information makes it relatively simple to use the periodic table to obtain electron configurations, as the following example shows.
Obtain the electron configuration for (a) Nb; (b) Pr.
Solution
a) Nb, element number 41, is found in the fifth period and in a region of the periodic table where a d subshell is filling (the second transition series). Moving backward (toward lower atomic numbers) through the periodic table, the nearest noble gas is Kr, and so we use the Kr kernel:
Nb [Kr] _____
The next element after 36K is 37Rb in which the 5s subshell is filling. Moving right one more space, we come to 38Sr which has a 5s2 pair. So far we have Nb [Kr] _____ 5s2 for the Nb configuration. We now move farther right into the 4d subshell region of the periodic table and count over three spaces (Y, Zr, Nb) to reach Nb. The total electron configuration is thus Nb [Kr]4d35s2 (Note that the principal quantum number of the d subshell is 4 ― one less than the number of the period. Also, if you look at the table of electron configurations, it should be noted that Nb is an exception to the typical orbital filling rules) b) A similar procedure is followed for Pr, element number 59. Moving backward through the table, the nearest noble gas is Xe, and so we use the Xe kernel. Counting forward again, Cs and Ba correspond to 6s2. Then La, Ce, and Pr correspond to three more electrons in the 4f subshell. The configuration is thus Pr...[Xe]4f36s2One more point needs to be emphasized about the relationship between electron configuration and the periodic table. The atoms of elements in the same vertical column of the table have similar electron configurations. For example, consider the alkaline-earth elements (group IIA). Using our rules for deriving electron configurations (Example 1) we have
| Element | Electron Configuration | Lewis Diagram |
|---|---|---|
| Be | [He]2s2 | Be: |
| Mg | [Ne]3s2 | Mg: |
| Ca | [Ar]4s2 | Ca: |
| Sr | [Kr]5s2 | Sr: |
| Ba | [Xe]6s2 | Ba: |
| Ra | [Rn]7s2 | Ra: |
Thus the similarities of chemical behavior and valence noted earlier for these elements correlate with the similarities of their outermost electron clouds. Such similarities account for the success of Mendeleev’s predictions of the properties of undiscovered elements.


