E1. Introduction to Drug Development
- Page ID
- 132806
Typically, drugs have been found by an almost serendipitous process. Active molecules are usually derived from biological sources such as bacteria, fungi, plants, etc. Different molecular species are extracted which, in bioassays, seem to inhibit a target biological function or specific enzyme. The structure is then solved, and medicinal chemists "decorate" the molecule by covalently modifying the initial structure in ways which will improve its medicinal value (decreased Kd, increased solubility, longer half-life, fewer side effects, etc.) Bioactive structures are often found to be peptides or modified peptides. One main job of a medicinal chemists is to synthesize a peptidomimetic with different functional groups than are found in peptides, making the structure more stable to endogenous proteases. Newer methods involve molecular modeling and rational drug design. Computers can be used to analyze the structure and activity of a large number of different molecules which inhibit the target enzyme. By doing Quantitative Structure Activity Result studies (QSAR), the computer can be used to hypothesize the structure of the "perfect" inhibitor, which can then be synthesized. In an analogous fashion, the active site of the target enzyme can be constructed from this data, without the benefit of having a crystal and NMR structure.
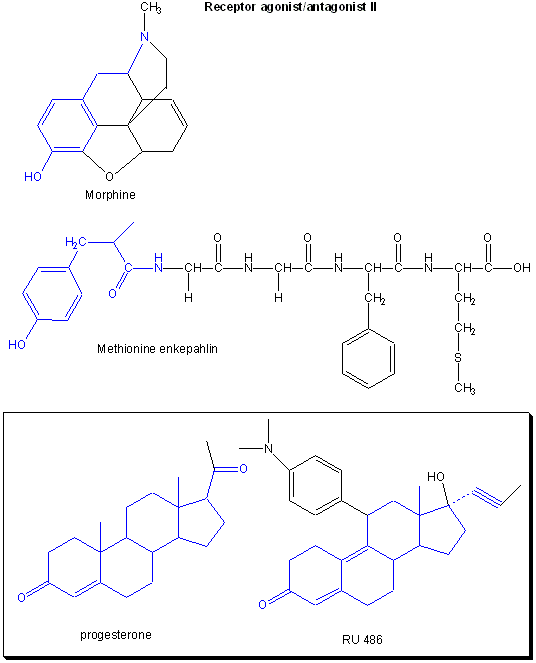
Drugs can be designed to inhibit enzymes or to bind to sites on receptors (either in the membrane or the cytoplasm). When bound to a receptor, the drug ligand may mimic the natural ligand and lead to expression of the biological activity of the receptor. In this case, the drug is an called an agonist of the normal ligand. In general, if the drug inhibits the activity of the bound receptor, the drug is called an antagonist. Examples are show below. Notice the similarity and differences in structures between the normal ligand and the drug. A more detailed description of agonist and antagonist will be present later in the discussion of enzyme inhibitors.
Figure: Receptor agonists/antagonist I
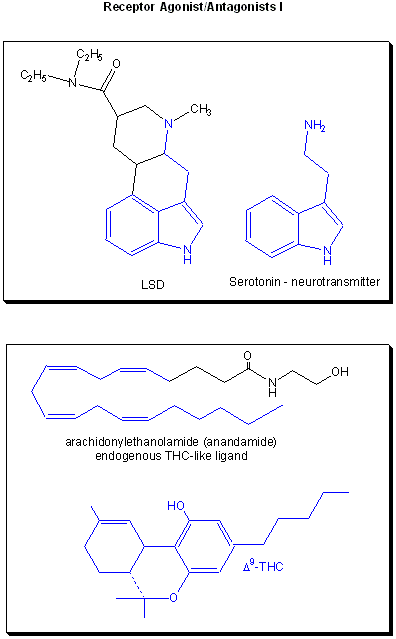
Figure: Receptor agonists/antagonist II