Diffraction and Bragg’s Law
- Page ID
- 78103
Take a look at the diagram below:
- When X-rays interact with a single particle, it scatters the incident beam uniformly in all directions.
- When X-rays interact with a solid material the scattered beams can add together in a few directions and reinforce each other to yield diffraction. The regularity of the material is responsible for the diffraction of the beams.
You have likely seen diffraction before. For example if you look at a CD when exposed to white light you can see it diffracted into varies wavelengths of color. The pits (or grooves) in the CD are the regularity of the material that causes the diffraction.

Google images: outreach.atnf.csiro.au
Diffraction can occur when any electromagnetic radiation interacts with a periodic structure. The repeat distance of the periodic structure must be about the same wavelength of the radiation. For example, light can be diffracted by a grating having scribed lines arranged on the order of the wavelength of light.
So if all electromagnetic radiation can diffract, why are X-rays used in crystallography?
X-ray Diffraction and Bragg’s Law
X-rays have wavelengths on the order of a few angstroms (1 Angstrom = 0.1 nm). This is the typical inter-atomic distance in crystalline solids, making X-rays the correct order of magnitude for diffraction of atoms of crystalline materials.
How are Diffractions Patterns Made?
When X-rays are scattered from a crystalline solid they can constructively interfere, producing a diffracted beam. What does this mean?
Constructive vs. Destructive Interference
Interference occurs among the waves scattered by the atoms when crystalline solids are exposed to X-rays. There are two types of interference depending on how the waves overlap one another.
Constructive interference occurs when the waves are moving in phase with each other. Destructive interference occurs when the waves are out of phase.
This constructive interference results in diffraction patterns.
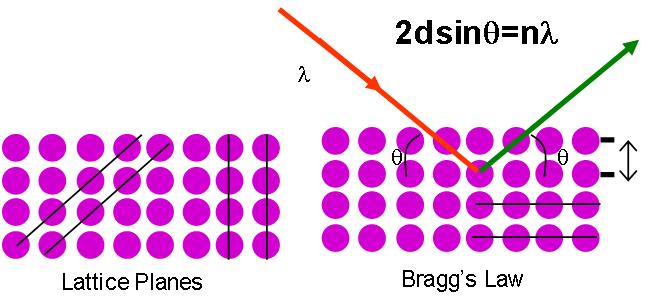
Bragg's Law and Diffraction
The relationship describing the angle at which a beam of X-rays of a particular wavelength diffracts from a crystalline surface was discovered by Sir William H. Bragg and Sir W. Lawrence Bragg and is known as Bragg’s Law
\[\mathrm{2d\sin θ= nλ}\]
λ = wavelength of the x-ray
θ = scattering angle
n = integer representing the order of the diffraction peak.
d = inter-plane distance of (i.e atoms, ions, molecules)
Click on the following image below to get to an Applet where you can explore this relationship of Bragg’s Law
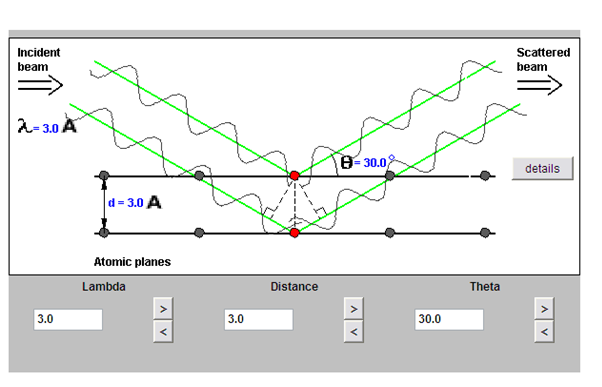
www.eserc.stonybrook.edu/ProjectJava/Bragg/
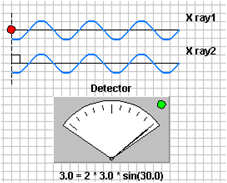
Guide to how to use Applet: There are 2 rays incident on two atomic layers of a crystal (d). At the beginning the scattered rays are in phase and interfering constructively. Bragg’s Law is satisfied and diffraction is occurring. If you click on the details button you can see the detector, which measures how well the phases of the two rays match. When the meter is green it indicates that Bragg’s law is satisfied. You can change three variables (d, λ, and θ) to see how they effect the diffraction.
The diffraction pattern of copper metal was measured with X-ray radiation of wavelength of 1.315Å. The first order Bragg diffraction peak was found at an angle 2theta of 50.5 degrees. Calculate the spacing between the diffracting planes in the copper metal.


