Part I. The Analytical Problem
- Page ID
- 227262
Investigation 1: Properties of Danshen's Constituent Compounds
The dried root of Salvia miltiorrhiza—also known as red sage, Chinese sage, or Danshen, where "dan" and "shen" are Chinese for "red-colored" and "tonic herb," respectively—is a traditional Chinese herbal medicine used to treat a variety of cardiovascular and cerebrovascular diseases, presumably because of its ability to prevent the formation of blood clots and its ability to dilate blood vessels [1]. Danshen is widely available throughout China, and is available, although to a lesser extent, in Europe and in the United States. The drug Dantonic®, a formulation that includes Danshen, is approved in 26 countries for the treatment of and prevention of angina; it currently is in phase III testing for use in the United States [2].
As with any medicinal plant, the chemical composition of Danshen is complex with more than 70 constituent compounds identified in the literature. Early studies of Danshen's chemical composition focused on lipophilic molecules, the four most important examples of which are:
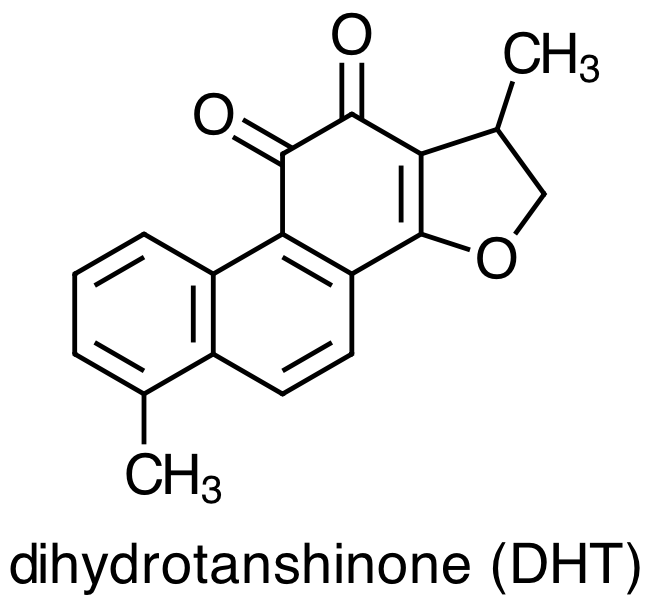
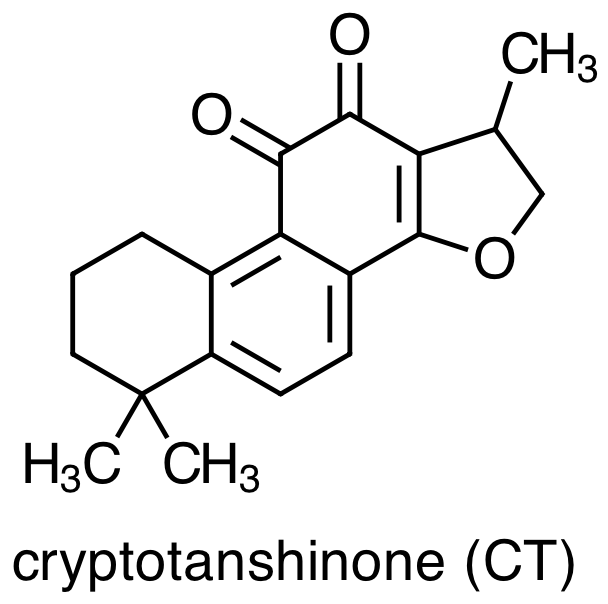
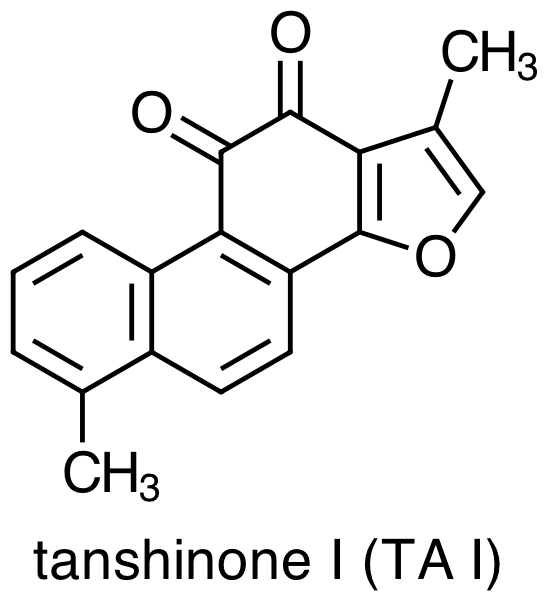
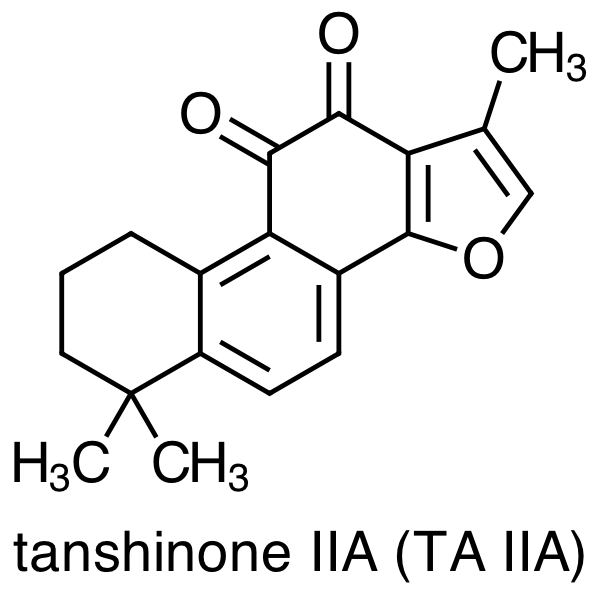
Danshen also contains hydrophilic constituents, the four main examples of which are:
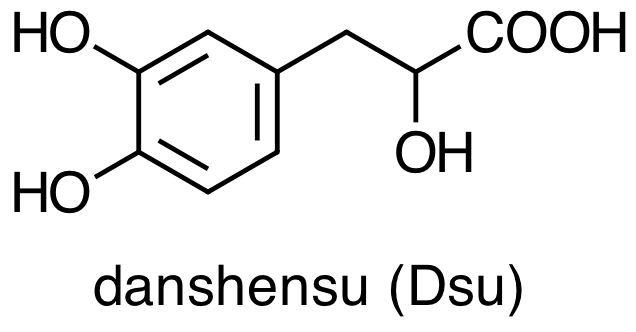
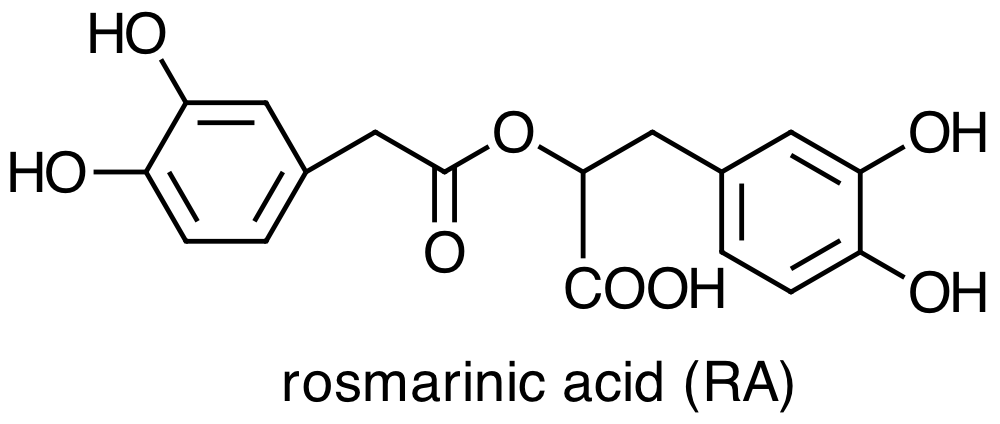
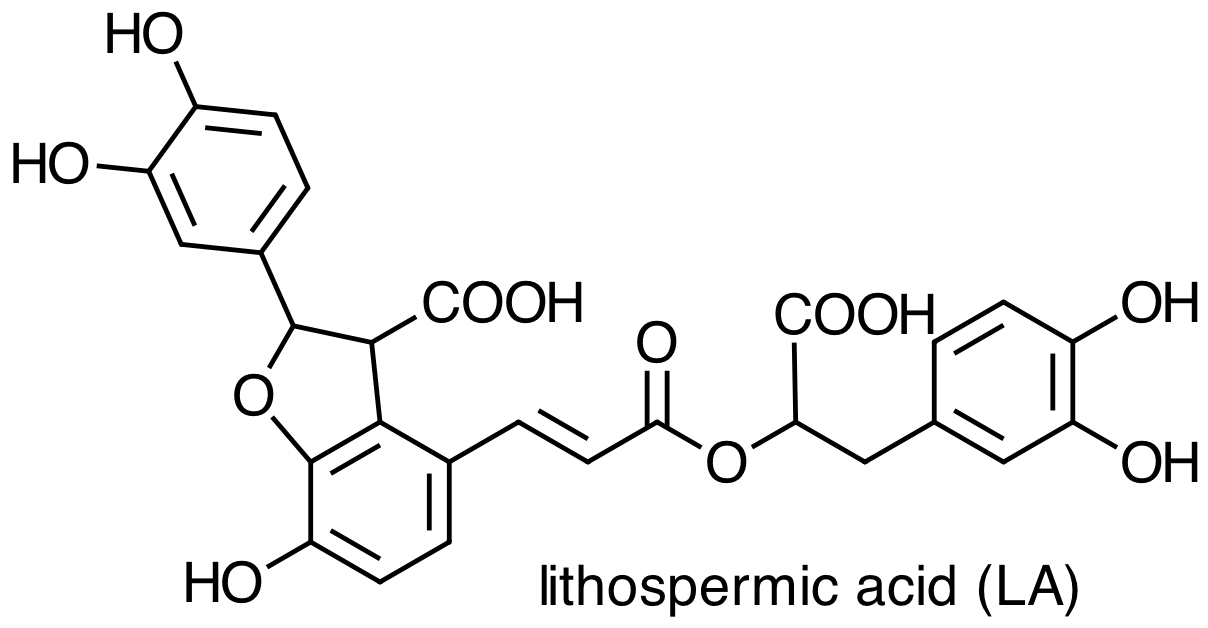
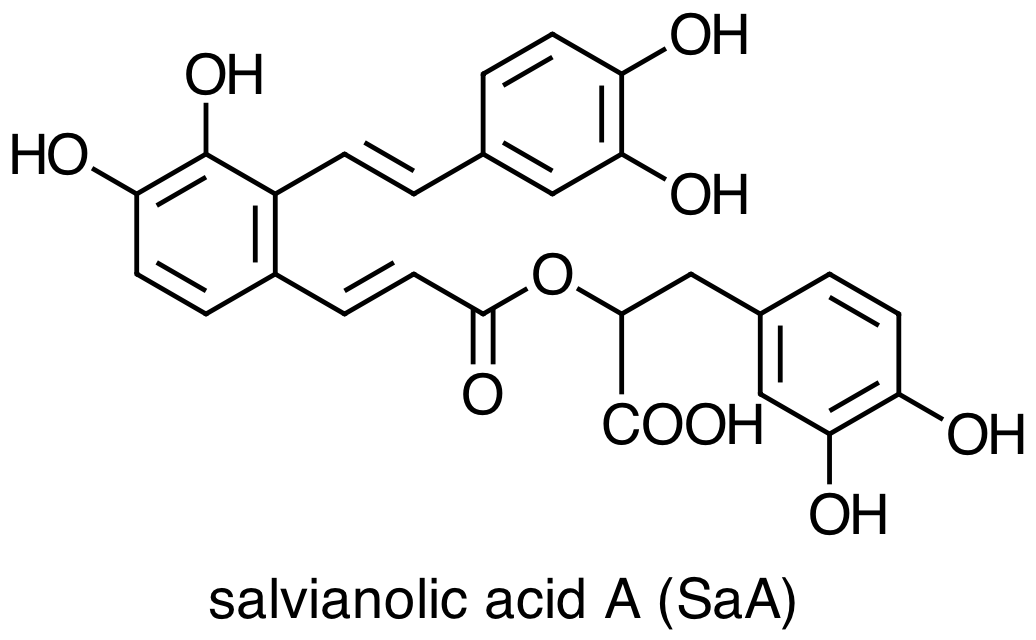
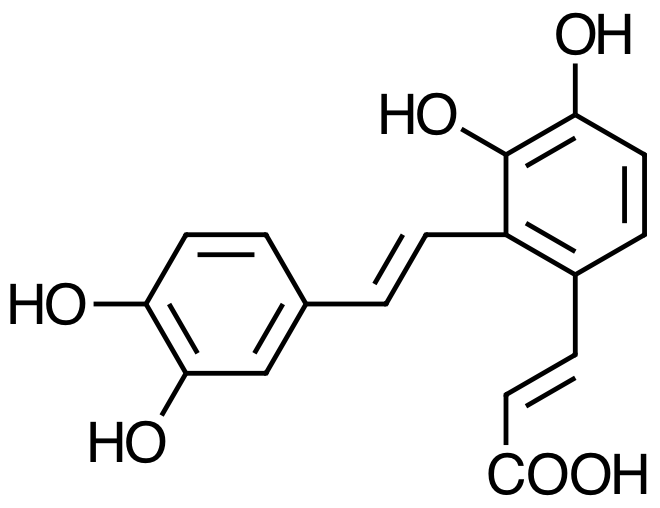
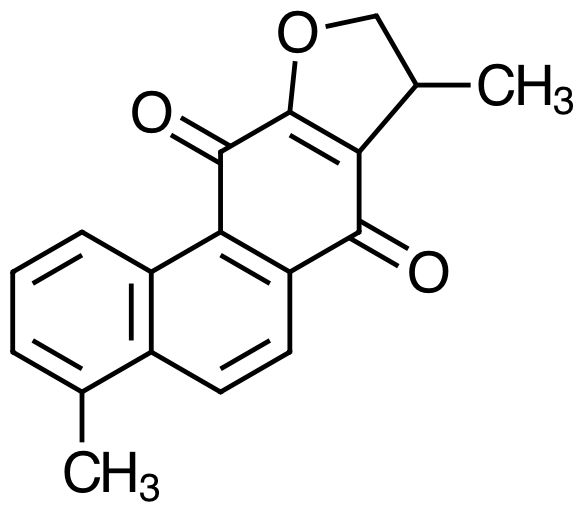
What does it mean to characterize a molecule as hydrophilic or as lipophilic? How do they differ in terms of their chemical or physical properties [3]? Are there structural differences between these two groups of molecules that you can use to classify them as hydrophilic or as lipophilic? Consider the molecules below, both minor constituents of Danshen, and classify each molecule as lipophilic or hydrophilic.
[1] For a review of Danshen’s medicinal properties and uses, see “Danshen: An Overview of Its Chemistry, Pharmacology, Pharmacokinetics, and Clinical Uses,” the full reference for which is Zhou, L.; Zuo, Z.; Chow, M. S. S. J. Clin. Pharmacol. 2005, 45, 1345-1359 (DOI).
[2] You can view details regarding the phase III trial at clinicaltrials.gov; the estimated completion date for the study is December 2016.
[3] A useful resource for exploring the chemical and physical properties of molecules is the Royal Society of Chemistry’s ChemSpider, a free database that provides access to the properties of over 30 million compounds.


