Electrochemistry
- Page ID
- 283142
CHEM 2300, In-class Exercises, Class 25
Name: _________________________
- Write the relevant half reactions and calculate Eo and K for the following reaction. Is the reaction spontaneous as written?
\[\ce{I2(s) + 5 Br2(aq) + 6 H2O ↔ 2IO3- + 10Br- + 12H+}\nonumber\]
\[\mathrm{E^o_{I_2} = 1.210}\nonumber\]
\[\mathrm{E^o_{Br_2} = 1.098}\nonumber\]
- Write the line notation for a cell made up of the following half reactions if the indicator electrodes are platinum and a monoprotic acid is in solution in the cathode cell. If a balanced reaction is not given, assume that the larger Eo value is the cathode.
\[\ce{Fe^3+ + e- ↔ Fe^2+} \hspace{50px} \mathrm{E^0 = 0.771}\nonumber\]
\[\ce{Cr2O7^2- + 14H+ +6e- ↔ 2Cr^3+ + 7H2O} \hspace{30px} \mathrm{E^0 = 1.36}\nonumber\]
- Given the following reaction, assign oxidation states to each element and write the 2 half reactions (both as reductions).
\[\ce{2 AgCl(s) + Pb(s) + 2F- (aq) ↔ 2 Ag(s) + 2Cl- (aq) + PbF2(s)}\nonumber\]
- Write the line notation for this cell.
- If the concentrations of NaF and KCl are both 0.10 M, calculate the cell potential for the cell made up of the above half reactions at 25 oC:
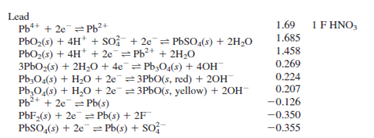
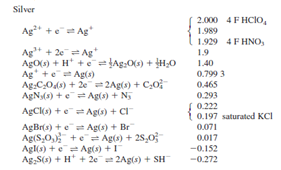
CHEM 2300, In-class Exercises, Class 26
Name: _________________________
- The apparatus in the figure below was used to monitor the titration of 50.0 mL of 0.100 M AgNO3 with 0.200 M NaBr. Calculate the cell voltage at each volume of NaBr, below.
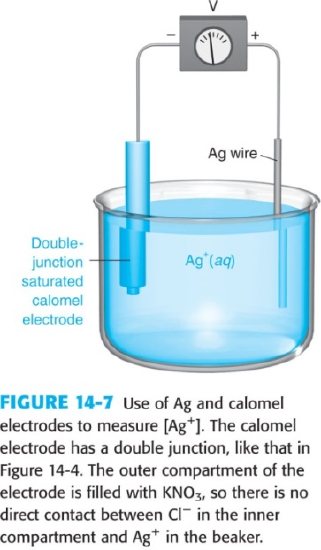
\[\mathrm{K_{sp}\: for\: AgBr(s) = 5.0 \times 10^{-13}}\nonumber\]
\[\mathrm{E_{S.C.E.}=0.241\: V}\nonumber\]
- 1.00 mL
- 25.1
- Write the half reaction(s) for the following reactions, as reductions:
- Pt (s) | Cr3+ (0.10 M), Cr2+ (0.050 M)
- Cd(s) | Cd2+ (1 M) || H+(aq, 1 M), Mn2+ (l M), MnO4- (l M) | Pt
- Cr2O72-(aq), Cr3+(aq), HA(aq) |Pt(s)
- Pt (s) | Cr3+ (0.10 M), Cr2+ (0.050 M)
- A solution contains 0.100 M Ce3+, 1.00x10-4 M Ce4+, 1.00x10-4 M Mn2+, 0.100 M MnO4-, and 1.00M HClO4.
- Write a balanced net reaction that can occur between species in this solution, assuming the Ce half cell is the cathode.
- If Ecell is measured at -0.019 V, calculate the equilibrium constant for the balanced net reaction.
\[\mathrm{K=10^{n*E^o/0.05916}}\nonumber\]
Contributors and Attributions
- Sarah Gray, Stockton University (sarah.gray@stockton.du)
- Sourced from the Analytical Sciences Digital Library


