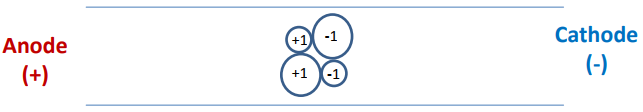Name: ______________________________
Section: _____________________________
Student ID#:__________________________
Work in groups on these problems. You should try to answer the questions without referring to your textbook. If you get stuck, try asking another group for help.
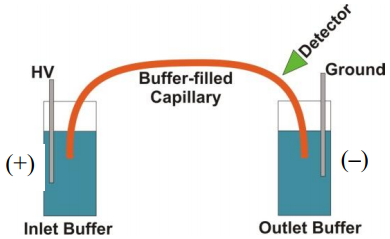
- What is the functional group on the surface of a fused silica capillary column? Fused silica is a silicon dioxide (SiO2) polymeric material.
- Would the nature of the interior surface of the capillary be different if the background electrolyte was at pH 2 versus pH 9?
- Assuming the silanol groups are deprotonated to anionic silanate groups, what do you think will happen in the portion of the solution in immediate contact with the surface of the fused silica?
- What happens in the diffuse layer in the presence of this applied electric field?
- Which flow profile, hydrodynamic or electroosmotic, results in greater peak broadening?
- As previously discussed, broadening in chromatographic columns results from eddy diffusion, longitudinal diffusion, and mass transport broadening in the stationary and mobile phases. Which of these processes contribute to peak broadening in capillary electrophoresis?
- What would happen to the electroosmotic flow rate at pH 2.5 versus pH 10?
- Suppose the compounds being analyzed have a charge.Is there any other process that will result in analyte ions moving in the capillary column?
- What variables influence the electrophoretic migration rate of a charged analyte?
- Imagine that four ions start in the middle of the capillary as shown below. Assuming that the ions only move as a result of electrophoresis, draw the position of the ions after 30 kV has been applied for a short time.
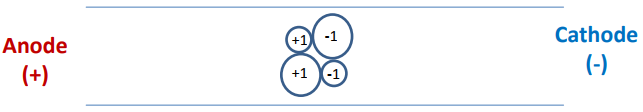
- Now consider the separation of a sample that has anionic, cationic and neutral analytes and has both electroosmotic and electrophoretic flow. The detector is placed on the end of the capillary near the cathode as shown below. Anions, cations and neutrals are all detected. Explain.

- What is the predicted elution order of the following five substances? In addition, draw an electropherogram of the results and explain your reasoning.
Substance A: neutral
Substance B: ̶ 1 charge
Substance C: +1 charge
Substance D: neutral
Substance E: ̶ 2 charge
Substances A, B, C, and E are all the same size. Substance D is twice as large as the others.
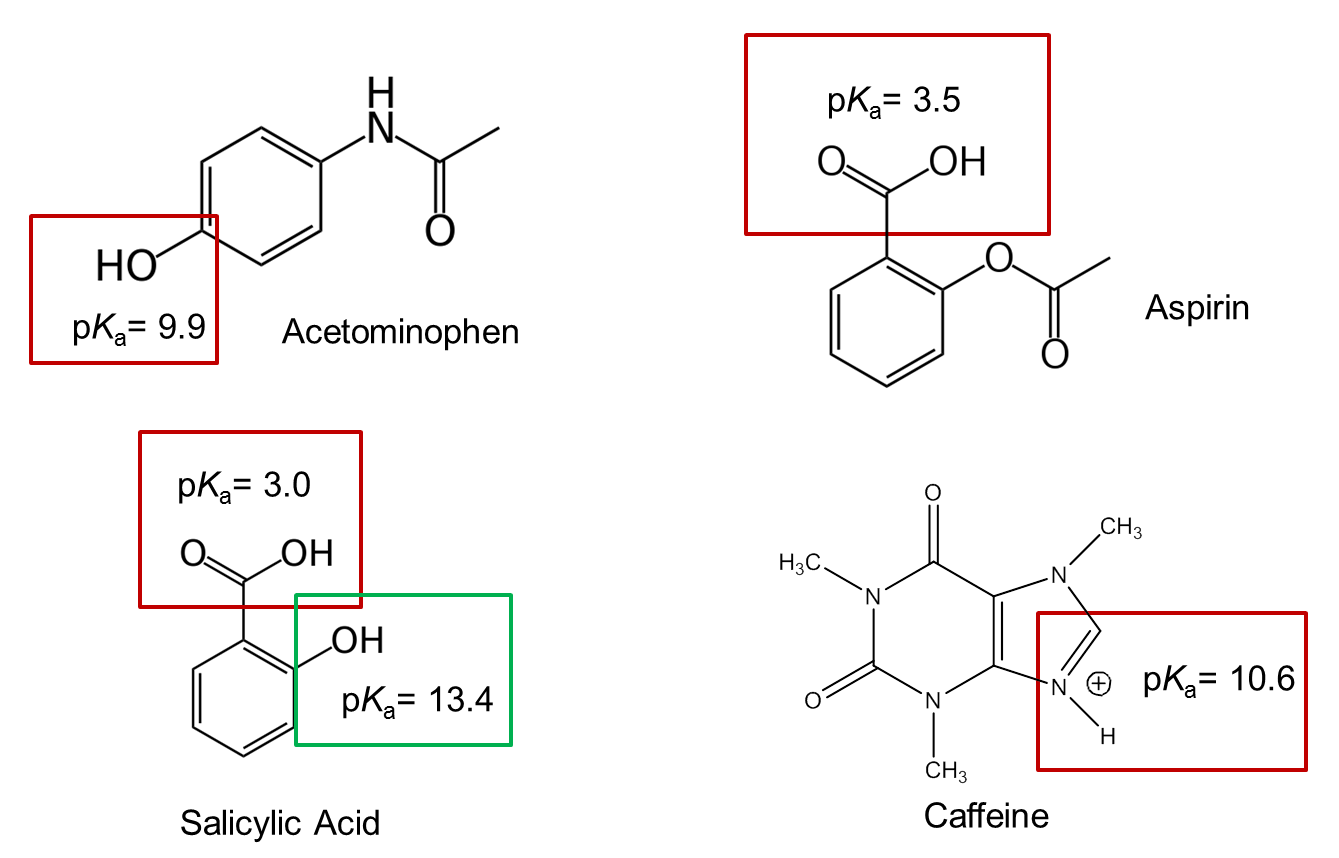
- The structural formulas and relevant pKa values for the four components of an Exedrin tablet are shown below. (The protonated form of the boxed group is shown.) Predict the elution order in CE with a 20 mM borate buffer at pH = 9.3.
- What could you do experimentally to confirm your predicted elution order?