19.1: Information Contained in Proton NMR Spectra
- Page ID
- 221078
1. NUMBER OF SIGNALS. The number of signal present in an NMR spectrum reflects the number of magnetically different protons. For our purposes, although not alwyas true, we will assum that magnetically different protons are also chemically different. Thus the number of signals reflects the number of chemically different protons or sets of protons. For example methanol has two different sets of protons (methyl and hydroxyl) and therefore is expected to show two signals in the proton NMR spectrum.
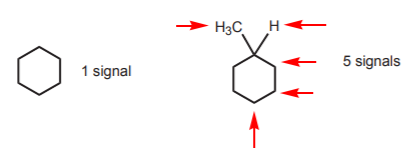
There is a direct relationship between the symmetry of the molecule and the number of proton NMR signals it exhibits. The following example illustrates this point dramatically. The cyclohexane molecule exhibits only one signal at room temperature. The mere introduction of a methyl group changes the spectrum to five signals, corresponding to 5 different sets of protons.
2. RELATIVE INTENSITIES OF THE PEAKS, OR SIGNALS. The term intensity, when used in reference to NMR signals, indicates the area under the peak. The areas under the peaks are given by an integrator when recording a spectrum. The relative intensities of the signals reflect the relative numbers of protons of different kinds present in the molecule. The following examples illustrate this point.

3. CHEMICAL SHIFTS. The relative positions of the signals along the frequency axis in the δ-scale is a reflection of the extent of electron density around the different types of protons. See the Introduction to NMR manuscript for the explanation of relevant terms such as shielded, deshielded, etc. Also, consult a table of chemical shifts typical of proton signals for specific functional groups. In your laboratory textbook, refer to appendix 4, pages A37 to A40.
4. SIGNAL MULTIPLICITY AND SPIN-SPIN SPLITTING. Each signal in a proton NMR spectrum may or may not be split into one or more peaks. This is called signal multiplicity and gives rise to names such as singlet, doublet, triplet, quartet, pentet, and multiplet. The most common concept associated with signal multiplicity is the n+1 rule. According to this rule, the signal for the proton under observation will be split into n+1 peaks by protons attached to adjacent carbons, where n is the number of such protons.
Another concept associated with signal splitting is referred to as “complex splitting.” When the NMR signals of certain protons do not follow the n+1 rule they are said to exhibit complex splitting. An understanding of coupling constants, to be discussed next, enables an understanding of complex splitting and the n+1 rule limitations. Section NMR.7 on page A40 of the lab textbook contains a brief discussion of the n+1 rule.
5. COUPLING CONSTANTS. When the proton under observation is split into several peaks by neighboring protons, the distance between these peaks is a constant called the coupling constant (J). Any two sets of protons that exhibit the same coupling constant are most likely spltting each other’s signals and are said to be coupled. This bit of information can be very helpful when assigning NMR signals to specific sets of protons in a molecule. A table showing typical coupling constant values for common types of protons is shown on page A43 of the lab textbook.


