15.1: The Versatile Nature of the Carbonyl Group
- Page ID
- 213821
Like alcohols, the carbonyl group is a reactivity package concentrated in a small structure. Examination of this structure through its resonance forms reveals unshared electron pairs, which give the molecule basic or nucleophilic properties. It also reveals an electron-deficient carbon, which gives the molecule electrophilic character. Finally, the a-hydrogens possess acidic character by virtue of their being close to the electrophilic carbon. The easiest way to see this is by realizing that the conjugate base is resonance-stabilized.
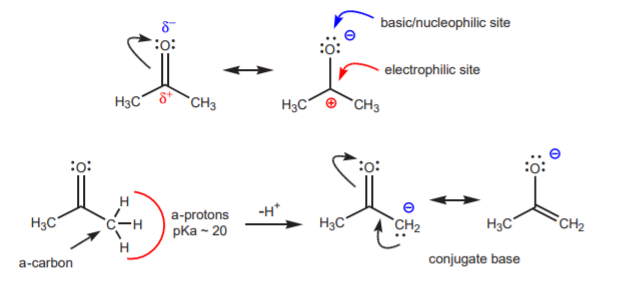
The conjugate base shown above is an enolate ion, so called because it is also the conjugate base or an enol. An enol is a functional group that contains an alkene (C=C bond) and an alcohol (hydroxyl group) that share a common carbon.
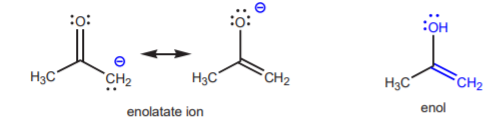
The enolate ion itself has basic/nucleophilic properties. Although the oxygen and the a-carbon share the negative charge, periodic trends indicate that carbon is more nucleophilic than oxygen. A negative charge on the more electronegative oxygen is more stable (less reactive) than a negative charge on the less electronegative carbon (more reactive). When the enolate ion reacts with a typical electrophile such as methyl iodide it undergoes C-alkylation rather than O-alkylation. Another name for this reaction is a-alkylation.
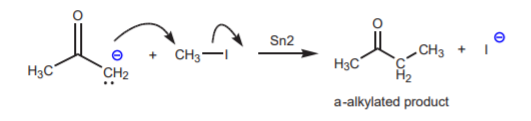
Although this is a very useful reaction, it suffers from a number of drawbacks, as discussed in the lab textbook, p. 348 - 349. Some of these drawbacks include polyalkylations, side reactions, and mixtures of products.
A better alternative is to perform the reaction by means of an enamine. Enamines are functional groups that contain an alkene (C=C bond) and an amine (sp3 nitrogen) that share a common carbon. Like the enolate ion, the enamine also has resonance structures that reveal the nucleophilic character of the a-carbon.
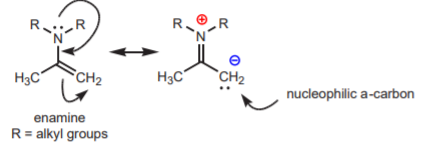
The enamine undergoes \(\alpha\)-alkylation in a manner analogous to that shown for the enolate ion, but with less problems.
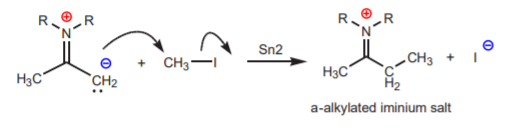
The iminium salt can be easily transformed into the corresponding carbonyl group through acid-hydrolysis. This is a reversible reaction, which means that a carbonyl compound can be transformed into an enamine by treatment with a secondary amine in acid medium as well. Secondary amines are preferred over primary amines because they only have one N-H bond. Once the N-H hydrogen is used in the first step, it is no longer needed. A primary amine has two N-H hydrogens and the second one would cause problems during subsequent steps.
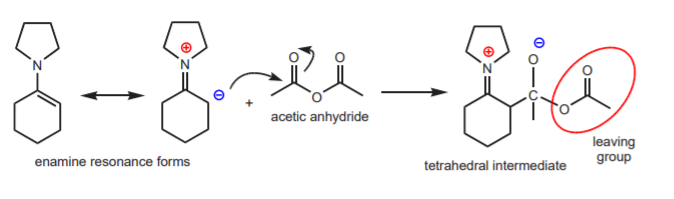
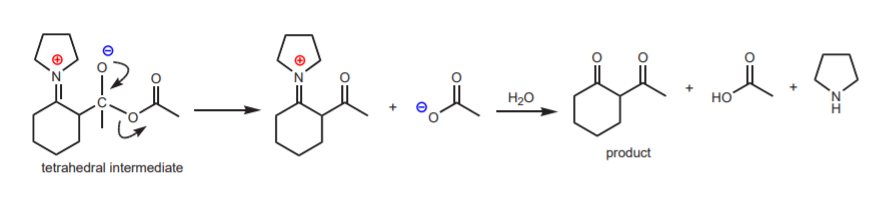
Some of the most commonly used secondary amines are cyclic ones, such as pyrrolidine. The complete reaction sequence, from carbonyl compound to a-alkylated carbonyl compound via enamines is shown below, using cyclohexanone as the carbonyl compound and pyrrolidine.
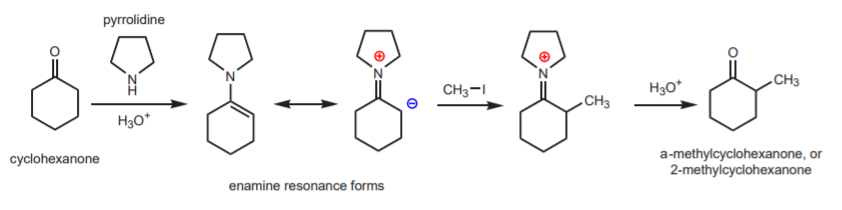
These are exactly the same substances that will be used in experiment 43, except that the reaction will be an acylation, rather than an alkylation. The acylating agent will be acetic anhydride. The acid used in the first step will be the strong organic acid p-toluenesulfonic acid. These two substances are strong irritants and the acid is corrosive. Please handle everything in the hood with gloves.
The following mechanisms show the acylation steps.