9.1: Grignard Reactions- Theory and Background
- Page ID
- 213812
NATURAL SOURCES OF ORGANIC CHEMICALS: All forms of life, dead or alive, such as animals, plants, and fossils.
OIL (fossil fuels): Made up primarily of hydrocarbons, notably alkanes, alkenes, and aromatics (benzene-based compounds).
ALKANE FUNCTIONALIZATION: The intent is to introduce a reactive group (FG1) in the otherwise unreactive alkane chain. Once a reactive group is in place, other functional groups (FGn) can be introduced.
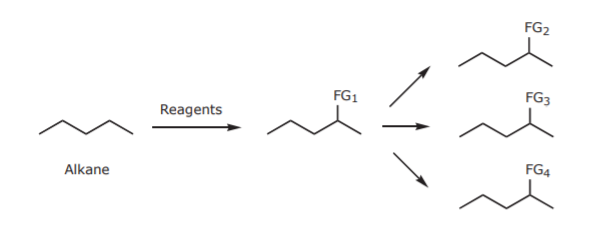
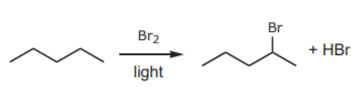
The choices for FG1 are very limited, especially in a laboratory setting. One of the few synthetically useful reactions that alkanes undergo is free radical halogenation, i.e. insertion of a halogen (normally chlorine or bromine) onto the carbon chain:
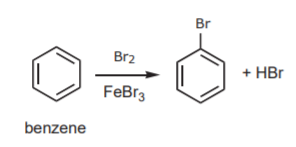
Aromatic hydrocarbons (benzene based) can undergo a modified version of this reaction:
Alkyl halides can be transformed into a variety of other functional groups by Sn2 reactions and eliminations:
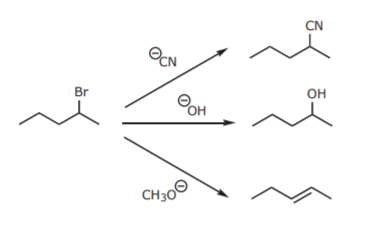
Alkyl halides can be converted into CARBON NUCLEOPHILES. Carbon nucleophiles enable chain expansion by formation of carbon-carbon bonds. Carbon nucleophiles contain a negatively charged carbon with an unshared electron pair. In most cases that happens when carbon is bonded to a metal.
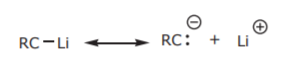
Such compounds are called organometallics.
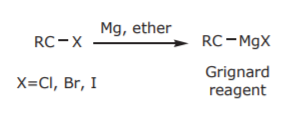
One of the most important and versatile classes of organometallics are those containing a carbon-magnesium bond. They are called GRIGNARD REAGENTS, after Victor Grignard, whose development of their chemistry won him a Nobel Prize. See his biography at http://www.nobel.se/chemistry/laurea...gnard-bio.html
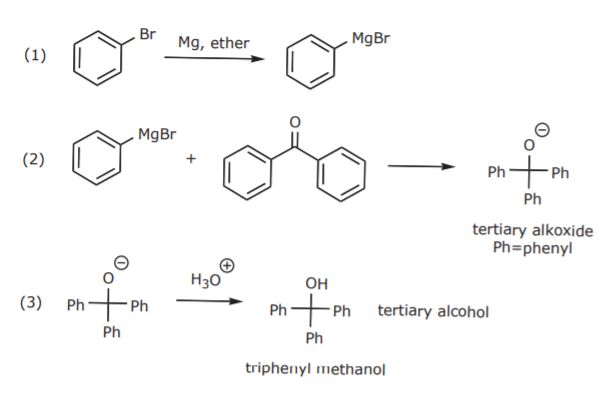
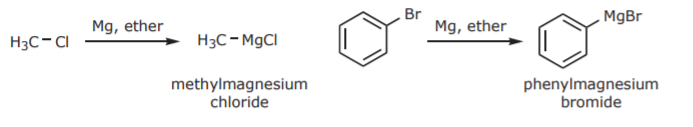
Grignard reagents are prepared by insertion of Mg metal into the carbon-halogen bond of alky and aryl halides:
Examples:
The corresponding resonance structures reveal the nucleophilic (and also basic) character of the carbon atom attached to Mg:
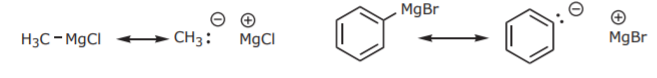
Their own synthesis makes it apparent that Grignard reagents are not very good nucleophiles for regular Sn2 reactions. If they were, they would attack the alkyl halide that’s used in their synthesis:
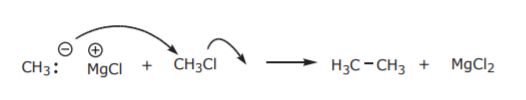
Instead, their main use is in nucleophilic addition reactions of carbonyl compounds. That is, substances containing C=O bonds. Such substances are usually aldehydes and ketones.
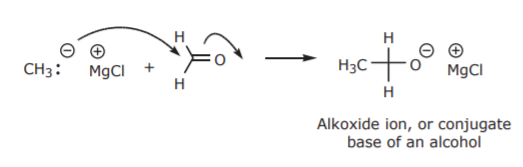
The alkoxide ion is not as stable as its conjugate acid, the alcohol. Therefore, this step is always followed by treatment with water or a strong acid to protonate the alkoxide and convert it into the more stable alcohol as the final product.
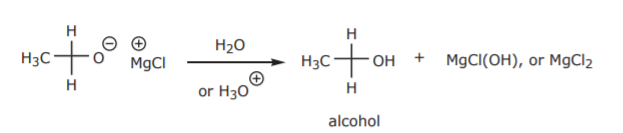
Most carbonyl compounds yield primary, secondary, or tertiary alcohols when reacted with Grignard reagents, depending on the specific case. The following table illustrates the synthetic range. For additional examples refer to your organic chemistry text.
GR + formaldehyde ----------> primary alcohol
GR + other aldehydes -------> secondary alcohol
GR + ketone -------------------> tertiary alcohol
Experiment 31A involves the preparation and use of phenylmagnesium bromide to prepare a tertiary alcohol by reacting the Grignard reagent with benzophenone (a ketone):