9.1: Oxidation and Reduction in Organic Chemistry
- Page ID
- 213347
The concepts of oxidation and reduction in chemistry are fundamentally related to the loss and gain of electrons, respectively. For a specific atom, oxidation brings about an increase in the oxidation number, whereas reduction does the opposite. This is of course true in organic reactions as well, but it is frequently less clear where the gain or loss of electrons is taking place. For this reason, the concepts of oxidation and reduction are addressed from a different perspective in organic chemistry.
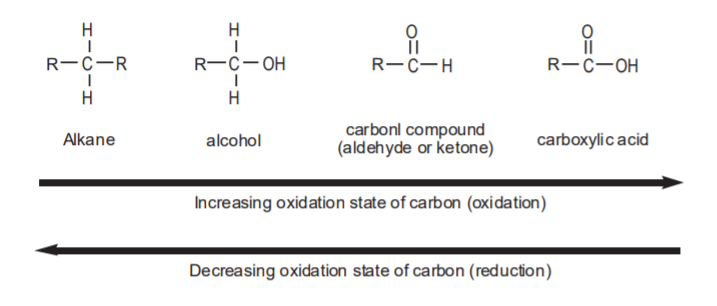
Since carbon is the most important element in organic chemistry, we can define oxidation as a process whereby carbon gains bonds to more electronegative atoms. These can be any atoms more electronegative than carbon, such as chlorine or sulphur, but the most commonly seen in organic oxidations is oxygen. The more bonds to oxygen, the higher the oxidation state of the carbon involved. Examples of oxidation reactions are those that convert alkanes into alcohols, or alcohols into carbonyl compounds. The functional groups that contain the most highly oxidized form of carbon in organic compounds are the carboxylic acids and their derivatives.
Reduction is the opposite of oxidation. We can define reduction as a process whereby carbon adds bonds to less electronegative atoms. Again, the most common of these atoms is hydrogen. Therefore, any reactions that cause carbon to add more bonds to hydrogen can be referred to as reductions. Examples are the conversion of carbonyl compounds into alcohols, or even alkanes. Obviously, alkanes represent the most reduced (least oxidized) type of organic compounds possible.
The following scheme shows the most common functional groups involved in organic oxidations and reductions. Any chemical conversions that proceed to the right can be classified as oxidations, and any reactions that proceed to the left can be classified as reductions.