6.3: Experiment 5- Instructions for Part B
- Page ID
- 212927
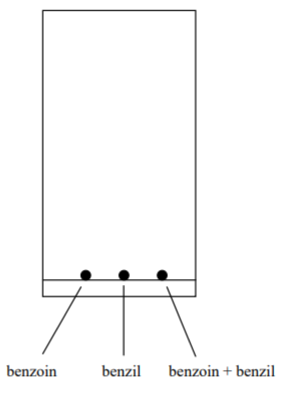
Although the lab textbook proposes several compounds and solvents that you can use in part B, we will use only two substances and three solvents. The two substances are benzoin and benzil. The solvents are hexane (nonpolar), methylene chloride (moderate polarity), and acetone (polar). Therefore, you will spot three solutions onto each of three TLC plates:
a. Benzoin only solution
b. Benzil only solution
c. Benzoin and benzil solution
1. PREPARE THREE TLC PLATES. Spot each plate with the above solutions, as follows:
2. DEVELOP THE 3 PLATES AS FOLLOWS:
• Plate 1 in hexanes
• Plate 2 in methylene chloride
• Plate 3 in acetone
3. CALCULATE Rf VALUES for the spots in the three plates.
4. REPORT RESULTS explaining which solvent effected the best separation of the benzoin-benzil mixture (third spot), and why, based on polarity of the substances and the solvents used.
The chemical structures of benzoin and benzil are given on p. 309 of the lab textbook, under Exp. 36B.


