1.4: Examples of Portions of Lab Reports
- Page ID
- 212210
EXAMPLE 1: INTRODUCTION TO A REPORT OF A PHYSICAL OPERATION
In this example, the students will perform a physical operation known as crystallization. This operation is intended to purify solids containing small amounts of impurities.
INTRODUCTION. In this experiment acetanilide will be purified by crystallization from water. The learning objectives are to learn the applications of this technique and its execution, and to learn how to take a melting point. The expected outcome is to obtain a pure substance from an initially impure sample and to demonstrate its purity by recording its melting point.
TABLE OF PHYSICAL CONSTANTS
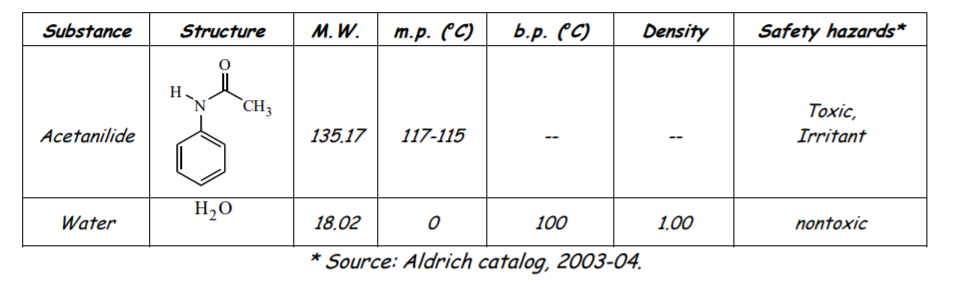
| Substance | Structure | M.W. | m.p. (°C) | b.p. (°C) | Density | Safety hazards* |
| Acetianilide |  |
135.17 | 117-115 | -- | -- | Toxic, irritant |
| Water | H2O | 18.02 | 0 | 100 | 1.00 | nontoxic |
* Source: Aldrich catalog, 2003-04.
No highly toxic substances are handled in this experiment. In case of skin contact with acetanilide, wash affected area with plenty of soap and water.
EXAMPLE 2: INTRODUCTION TO A REPORT OF A CHEMICAL REACTION, OR SYNTHESIS
In this example, the students will prepare 1-bromobutane from 1-butanol in an acid catalyzed reaction.
INTRODUCTION. In this experiment 1-bromobutane will be prepared from 1-butanol in an acid-catalyzed Sn2 reaction. The main learning objective is to illustrate a synthetic application of the Sn2 reaction. The expected outcome is to obtain a reasonably good yield of product with minimal formation of side products such as alkenes arising from elimination reactions.
CHEMICAL EQUATIONS
\(\: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \: \ce{CH3CH2CH2CH2OH + NaBr + H2SO4 -> CH3CH2CH2Br + NaHSO4 + H2O}\)
| 1-butanol | sodium bromide | sulfuric acid | 1-bromobutane | sodium bisulfate | |
| M.W. | 74.12 | 102.9 | 98.08 | 137.03 | |
| amounts used | .185g (185 mg) | .300g (300mg) | .250mL (.460g, or 460mg) | .342g (342mg) (theoretical) | |
| mmoles used | 2.50 | 2.92 | 4.69 | 2.5 (theoretical) |
CALCULATIONS
Calculating mass of sulfuric acid from its measured volume
\(Density = \frac{mass(g)}{volume(mL)} \rightarrow = density \times volume = 1.84 \frac{g}{mL} \times .25 mL = 0.46\)
Calculating mmoles from the mass of the substances
\(Moles = \frac{mass (g)}{MW (g/mol)}\)
Moles of 1-butanol \(= \frac{.185g}{74.12} = .0025moles = 2.5mmoles\)
Moles of NaBr \(= \frac{.300g}{102.9} = .00292g= 2.92mmoles\)
Moles of H2SO4 \(= \frac{.46g}{98.08} = .00469 moles = 4.69 mmoles\)
Calculating theoretical yield of product
The limiting reagent in this reaction is 1-butanol. The balanced equation shows that one mol of 1- bromobutane is obtained for every mol of 1-butanol used. Since we are starting with 2.5 mmoles of 1- butanol, then the theoretical yield of 1-bromobutane is also 2.5 mmoles, or .342 g.
You must note the following: (a) When handling very small amounts of substances it is more convenient to use millimoles (mmoles) instead of moles. (b) We are mainly concerned with reporting the properties of organic products such as 1-bromobutane, and not inorganic products such as sodium bisulfate. Inorganic products are typically separated and discarded. (c) Not all reagents are present in equimolar amounts. In such reactions, the limiting reagent is the one present in the smallest amounts, such as 1-butanol in this example.
TABLE OF PHYSICAL CONSTANTS
| Substance | Structure | M.W. | m.p (°C) | b.p (°C) | Density (g/mL) | Safety hazards* |
| 1-Butanol | CH3CH2CH2CH2OH | 74.12 | -89.8 | 117.5 | .81 | Flammable |
| Sodium bromide | NaBr | 103.9 | -- | -- | -- | Irritant |
| Sulfuric Acid | H2SO4 | 98.08 | -- | -- | 1.84 | Corrosive |
| 1-Bromobutane | CH3CH2CH2CH2Br | 137.03 | -112 | 100-104 | 1.276 | Flammable |
* Sources: Aldrich catalog, 2003-04, MSDS.
All substances must be handled in the hood, wearing gloves and eye protection. Avoid contact with eyes and inhalation. Skin contact procedures: Wash affected area with plenty of soap and water. If a skin rash or a burning sensation results, see a physician immediately. Eye contact with sulfuric acid requires immediate attention. Wash with plenty of water and see a physician as soon as possible.
REACTION MECHANISM
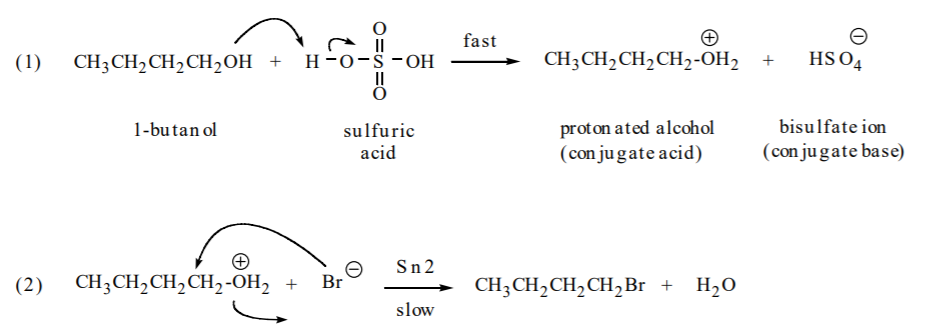
The reaction between sodium bromide and sulfuric acid generates HBr. There is a high concentration of bromide ions in this medium. The first step is a proton transfer to produce the protonated alcohol with a 7 good leaving group (water). In the second step, bromide ion acts as a nucleophile to displace the water and form the substitution product.
APPARATUS USED
A common setup such as a reflux or a distillation apparatus can be simply described with words, or a reference to another source can be given. More unique setups should be drawn to the best of the student’s ability. Here is an example:
A reflux setup such as the one shown on p. 198 of the Pavia textbook (4th ed.) was used. The only modification was the omission of the gas trap at the top, since this reaction was conducted in the hood.
EXAMPLE 3: EXPERIMENT OUTLINE and OBSERVATIONS
In this example, acetylglycine is being prepared by reaction of glycine with acetic anhydride. Here is what the experiment outline and corresponding observations might look like.
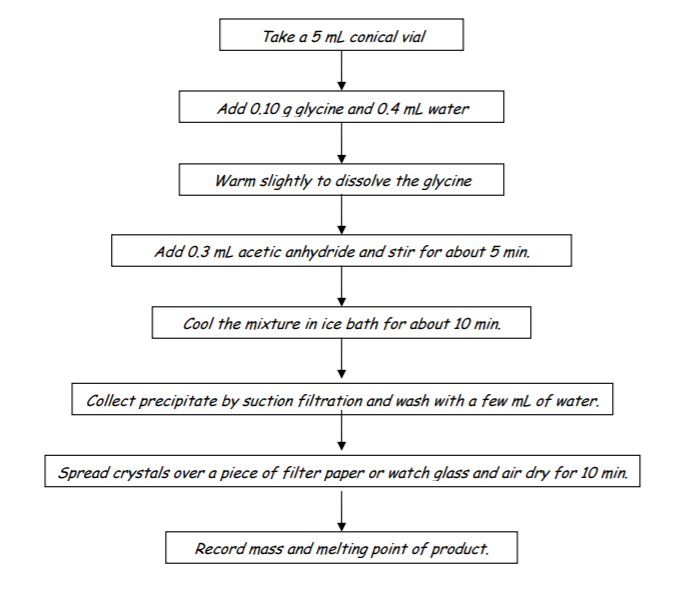
EXPERIMENT OUTLINE 1
1. Place 0.10 g glycine in a 5 mL conical vial
2. Add 0.4 mL water and warm slightly to dissolve
3. Add 0.3 mL acetic anhydride and stir mixture for about 5 min.
4. Cool mixture in ice bath for about 10 min.
5. Collect precipitate by suction filtration and wash with a few mL of water
6. Spread crystals over a piece of filter paper or watch glass and air dry for 10 min.
7. Record mass and melting point of product
OBSERVATIONS OF PHYSICAL CHANGES
a) When the glycine/water mixture was warmed, glycine dissolved after about 20 sec.
b) When the reaction mixture was cooled in ice, white, needle-shaped crystals formed.
c) After filtration and drying of the product, 0.14 g. of solid was obtained
d) The mp of the crystals was recorded as 205-207°
e) The solid product was possibly still slightly wet
1 See the next page for the equivalent flow chart of operations. Of course that’s a very simple flow chart. You might want to give it a different design, flow direction, or include pictures.
FLOW CHART OF OPERATIONS
EXAMPLE 4: RESULTS, DISCUSSION, AND CONCLUSIONS
In this example, impure acetanilide was purified by crystallization. The percent recovery and melting point were recorded.
RESULTS, DISCUSSION, AND CONCLUSIONS
The crystallization of acetanilide gave a 52 % recovery. Loss of product during the hot filtration accounts in part for the low recovery. The melting point of the crystallized product (110-112 oC) is higher than that of the crude material (105-109 oC), indicating a higher degree of purity. However, the melting point of the crystallized product is lower than the reported literature value (113-115 oC), indicating that it is not completely pure. Perhaps presence of residual water (crystallizing solvent) and error in the melting point determination can account for this. The conclusion is that the crystallized material was in fact acetanilide, based on its melting point, but it was not highly pure. A second crystallization should produce a fairly pure material, but it would also decrease the % recovery.


